|
Maxis Prepaid broadband. To use this service- 1Day-500MB-RM8 7day-2GB-RM25 How to get started? Step 1-Insert the Hotlink SIM into your mobile phone and make a call to activate it. Step 2-Call *100# and press SEND. Step 3-Select SERVICES and press SEND. Step 4-Select PREPAID BROADBAND and press SEND step 5-Choose a broadband package and wait for your confirmation SMS. Step 6-Insert the Hotlink SIM into a HSPA Modem or a HSPA enabled mobile phone and configure to the setting below: APN: unet Number to dial: *99# User ID: maxis Password: wap No additional configuration to the phone setting is required to use your mobile phone as a modem step 7-Start using the Internet via your browser _________________ now i confuse is the step 6 there.there write phone modem???????? And what- APN: unet Number to dial: *99# User ID: maxis Password: wap how to put the setting APN to unet? And 1 more i confuse is handphone got modem? But as i know my nokia 5130 XpressMusic there got write modem 56k. So i want ask you all my nokia 5130 XM can use this service?my phone got modem meh?how to do all this setting? Maxis Prepaid broadband. To use this service- 1Day-500MB-RM8 7day-2GB-RM25 How to get started? Step 1-Insert the Hotlink SIM into your mobile phone and make a call to activate it. Step 2-Call *100# and press SEND. Step 3-Select SERVICES and press SEND. Celcom Unlimited Data Plan at RM38 in Celcom, Malaysia Broadband, Mobile Operators 11 Comments 3,341 Views New Celcom Broadband plan at RM38 per month with max speed of 128Kbps, no contract. Step 4-Select PREPAID BROADBAND and press SEND step 5-Choose a broadband package and wait for your confirmation SMS. Step 6-Insert the Hotlink SIM into a HSPA Modem or a HSPA enabled mobile phone and configure to the setting below: APN: unet Number to dial: *99# User ID: maxis Password: wap No additional configuration to the phone setting is required to use your mobile phone as a modem step 7-Start using the Internet via your browser _________________ now i confuse is the step 6 there.there write phone modem???????? And what- APN: unet Number to dial: *99# User ID: maxis Password: wap how to put the setting APN to unet? And 1 more i confuse is handphone got modem? But as i know my nokia 5130 XpressMusic there got write modem 56k. So i want ask you all my nokia 5130 XM can use this service?my phone got modem meh?how to do all this setting? Celcom is currently experiencing serious network congestion. Since celcom broadband users about 420,000 users (not include daily unlimited, weekly unlimited, and pay per use user). That equal to 60% of the total market share. So i hope with the recent launch of digi broadband and the launching of maxis prepaid broadband will reduce celcom network congestion. As for now, i'll stick with my celcom broadband since i still satisfied with it. But from the survey i found that many celcom broadband user keep complaining lately. Celcom sama bumbung dengan Screamyx parent company jugak Which one do you prefer, a snail speed limited to 1GB per day OR a faster speed but limited to 500MB per day? Let's say a 1GB file will take Celcom 384kbps 5 hours to download but the same 1GB file will only take Maxis (someone mentioned 1000kbps? I'll based on that then) only 2 hours to download. For user which does not have that much leisure time, they will go for Maxis. I know I would prefer faster speed (for just rm2 extra, I don't mind paying). You can get more things done faster if you have fast connection. Same as you can get more things done if given a fast PC than a slow one. My point of view. Actually celcom use their own facility, not depend on streamyx. They have their own backbone as well as their own international gateway link. In my opinion, celcom bb performance is still acceptable at my area. Although not as fast as before. Some people prefer good speed, some people prefer consistency, some people prefer widest coverage, some people prefer cheap price. So its up to you. As for me, i dont need high speed. As long as i can do my work. And of course coverage is important because im travel a lot. Anyway, maxis early this year has announce they huge investment in improving their data services, and i believe they learn from mistake by over selling their maxis broadband before. Now celcom is the one who over sell their broadband. I got some internal information that celcom just add 4 new pipeline each with capacity 4Gb/sec in last april, and we all receive a good service but it only last till june. In July it become overcrowded again. Note: this morning im using celcom bb at KLCC and the speed is ok, but usually it will become slower in the afternoon. I got 2 people using celcom @ ipoh and speed is not good either one of them migrated over to maxis already and i now see big happy smile face - said when using celcom before even youtube need to wait for a lot of buffering. Well, i guess i might just stop there. This is a thread about 'Maxis Prepaid Broadband' not a brand-war or comparison of it's service with Celcom (in which I am positively sure there is another thread for this). And I suggest you should stop too epool86 - you sounds like a Celcom promoter! Actually celcom use their own facility, not depend on streamyx. They have their own backbone as well as their own international gateway link. In my opinion, celcom bb performance is still acceptable at my area. Although not as fast as before. Some people prefer good speed, some people prefer consistency, some people prefer widest coverage, some people prefer cheap price. So its up to you. As for me, i dont need high speed. As long as i can do my work. And of course coverage is important because im travel a lot. Anyway, maxis early this year has announce they huge investment in improving their data services, and i believe they learn from mistake by over selling their maxis broadband before. Now celcom is the one who over sell their broadband. I got some internal information that celcom just add 4 new pipeline each with capacity 4Gb/sec in last april, and we all receive a good service but it only last till june. In July it become overcrowded again. Note: this morning im using celcom bb at KLCC and the speed is ok, but usually it will become slower in the afternoon. Yes, that is the disadvantages of using celcom because it using shared IP. By the way, i never see that 'red thinggy message' like you mentioned. Like i said, maxis showing a lot of improvement after they improve their network capacity early this year. They learn from mistake, we need to balance the market share so each of service provider got fair market share so all of us can enjoy better service. So, if any celcom bb did not satisfied with the service, they can switch to maxis. I just want celcom broadband service come back to normal, so i can enjoy excellent service like before. Nowadays, during peak hour speed is slow. About stability/consistency. So far im satisfied, no major issue. This post has been edited by epool86: Sep 4 2009, 12:17 PM. Maxis recently having problem with their system. I subscribed weekly broadband and I over use 2Gb so wanna resubscribed again but cant so I called customer service twice and they still cant solve it. Luckily resubscribed again yesterday, maybe still new so might have that problem. Celcom last few weeks ago until now, my customers and I always got 'Service Currently Not Available' blue box message which quite annoying. We need to disconnect and connect only can use back the service but that problem will pop-up again every 5 minutes then we have to disconnect and connect again. I'm now very very happy with my maxis speed. Stable, fast and no network problem if compare to celcom. Depends what u purpose use for. For me my purpose is office work/msn/light youtube streaming/download 3-4 videos per week, ngam ngam 2G per week. But if extra 500mb for me, than enough for me already. Actually I recommed maxis coz is very stable compare to celcom. I'm using maxis and celcom broadband now. Celcom this 3 months I always get error from celcom. My many friends also get that message too. That makes me very headache. U can try to use 1 hour plan or 1 day plan la, see suitable for u or not. Sometimes go office, then dont need waste money subcribe for 1 month plan. Depends what u purpose use for. For me my purpose is office work/msn/light youtube streaming/download 3-4 videos per week, ngam ngam 2G per week. But if extra 500mb for me, than enough for me already. Actually I recommed maxis coz is very stable compare to celcom. I'm using maxis and celcom broadband now. Celcom this 3 months I always get error from celcom. 
My many friends also get that message too. That makes me very headache. U can try to use 1 hour plan or 1 day plan la, see suitable for u or not. For celcom, need to check 1st with them by calling their customer service.if still within their reinstate period.yes u can get back the number.need to reload rm10 with them. For tunetalk, u need to reload rm50. For maxis u need to kindly request back from them is u want it back in prepaid.otherwise just opt for postpaid then change back to prepaid for xox, need to call their customer service.no topup needed. Digi - NO re activate provided all the above option are only valid if the number are still within their reinstate period regardless of which telco that might differs on their reinstate period all the above are based on my real experience.
0 Comments

Payoneer debit card odesk Payoneer pre authorized transactions Payoneer card in odesk: Sitemap: Payoneer Prepaid Mastercard. Activate payoneer card. Payoneer: Login. 
You can activate your Payoneer Prepaid Card online or via the automatic phone activation service. To activate your card online: • Log in to your Payoneer • Make sure that the right card is selected in the drop-down menu at the top of the screen • Click on To view your status and activate your card Click Here! • Follow the on-screen instructions to activate your card. Please visit our to view step-by-step instructions. To activate your card on the phone: Call the phone activation service at 1-888-802-4442 (within the US) or 1-646-386-2392 (International). Please note: You cannot activate a card if it has not arrived yet. You will need the full details of the card in order to activate it.   
Carefully read this article to Activate Payoneer card. Payoneer like its PayPal counterpart is an online money transfer payment service. It is a quick and easy way to receive money worldwide, it is a registered member service provider (MSP) of the MasterCard incorporated. Payoneer services are unique and useful, it can be used to receive freelance earnings and affiliate commissions from various companies. Payoneer is a revolutionized online payment system, you can sign-up for free, and get your own prepaid MasterCard in 30 days or less. BENEFITS OF HAVING A PAYONEER MASTERCARD • With the Payoneer MasterCard, cash can be withdrawn using the ATM across the globe. • Funds can be received in as fast as 24 hours. • With a Payoneer card, online shopping is made possible. • As soon as your money hits your bank account, it is automatically converted to your local currency. • You have full access to your account and can check your account balance, and transactions made. Payoneer account holders can receive funds into their bank accounts or e-wallet, or thru a reloadable prepaid debit card (MasterCard), this can be used at point-of-purchase(POP). Payoneer gives customers the opportunity to possess a “Free MasterCard” (Debit Card) which is accepted worldwide for transactions and payments. STEPS TO ACTIVATE PAYONEER CARD. Activating the Payoneer card before using it, is very vital, that is why a step by step guide on how to activate Payoneer card is usually sent to the customer by Payoneer in the letter sent to them. Payoneer provides some alternatives for the activation of the Payoneer card which are; PHONE ACTIVATION: For USA area, the call center can be contacted at 1-888-802-4442 Payoneer. For those outside the USA, call the number at 1-847-461-1828 Payoneer, Ex-3 (International). ONLINE ACTIVATION • Go to • Click the”Activate your card” on the main page Payoneer. • Enter your username and password (ie) the username and password you used in registering Payoneer card • Click the”Activate Card”. • Create a pin (it must be a 4 digit pin) and click submit. • An email will be sent to you from Payoneer, which changes your status to active. After completing the pin change, your card is now active. Note; a card cannot be activated before it arrives (ie) you must receive the card in order for it to be activated because all the card details will be needed for the activation using a Payoneer card, simplifies the way you pay and get paid. Icewind Dale II Cheats For PC • Enable cheat codes To enable cheat codes, go to the game's configuration and enable the cheat option. Next, while playing, press [ctrl] + [tab] to open the console window. Then enter a cheat.  
Enable the cheat console using the Icewind Dale II configuration utility (under 'Game Settings' in the 'Game' tab), then. Mar 07, 2017 PC Cheats - Icewind Dale II: This page contains a list of cheats, codes, Easter eggs, tips, and other secrets for Icewind Dale II for PC.   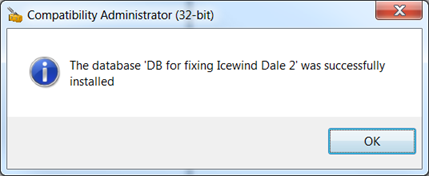 Hi, Thank you for posting this question on Microsoft Community Forums. I see that you want to know how to turn on cookies in Internet Explorer 10. 
How to enable Cookies in Internet Explorer. This guide will step you through the process of enabling Cookies in Internet Explorer. Select the 'Tools' menu. Jul 26, 2017. Learn how to view, manage, and delete cookies in Internet Explorer. Cookies can improve your browsing experience by allowing sites remember your preferences or letting you avoid signing in each time you visit certain sites. To block or allow cookies in Internet Explorer 11 for Windows 10. How to block or allow Cookies in Internet Explorer. Select the 'Tools' menu. In Internet Explorer 6, 7 or 8, 'Tools' is a menu at the top of the screen: Screenshot. In Internet Explorer 9 and higher, 'Tools' is found by clicking the gear icon on the right-hand side of the browser window: Screenshot showing the tools menu in. Let us work together to resolve this issue. Please answer these questions so that we can understand the issue better. Does the issue occur in Internet Explorer 10 desktop version or modern UI version? Do you have any anti-virus installed? What is the make and model of the tablet? Does the issue occur for a particular website? A cookie is a piece of text that a Web server can store on a user’s hard disk. When the same website is browsed again by the user in the future, the data stored in the cookie can be retrieved by the website to list the previous activities of the user. Follow these steps to enable cookies: a. Open Internet Explorer 10 desktop version. Click on the Tools button and select Internet Options. Click on Privacy tab and select Advanced.  
In the Advanced Privacy Settings window, select the settings you want and click on OK. Click OK again and restart Internet Explorer 10. Hope this information is useful. You can choose to delete existing cookies, allow or block all cookies, and set preferences for certain websites. What cookies are Cookies are files created by websites you visit. They make your online experience easier by saving browsing information. With cookies, sites can keep you signed in, remember your site preferences, and give you locally relevant content. There are two types of cookies: • First-party cookies are created by the site you visit. The site is shown in the address bar. • Third-party cookies are created by other sites. These sites own some of the content, like ads or images, that you see on the webpage you visit. Clear all cookies If you remove cookies, you'll be signed out of websites and your saved preferences could be deleted. • On your computer, open Chrome. • At the top right, click More Settings. • At the bottom, click Advanced. • Under 'Privacy and security,' click Content settings. • Click Cookies. • Under 'All cookies and site data,' click Remove all. • Confirm by clicking Clear all. Delete specific cookies. • On your computer, open Chrome. • At the top right, click More. • Click More tools Clear browsing data. • At the top, click the dropdown next to 'Clear the following items from.' • Choose a time period, such as the past hour or the past day. • Check 'Cookies and other site data.' Uncheck all the other items. • Click Clear browsing data. Change your cookie settings You can allow or block cookies saved by websites. Note: If you don't allow sites to save cookies, most sites that require you to sign in won't work. • On your computer, open Chrome. • At the top right, click More Settings. • At the bottom, click Advanced. • Under 'Privacy and security,' click Content settings. • Click Cookies. • Turn Allow sites to save and read cookie data on or off. Block cookies from other sites You can allow cookies from the site you visit, while blocking cookies from other sites that own ads or images on the webpage. To block these third-party cookies, turn on Block third-party cookies. All cookies and site data from other sites will be blocked, even if the site is allowed on your exceptions list. If you allow cookies by default, you can still block them for a certain site. • On your computer, open Chrome. • At the top right, click More Settings. • At the bottom, click Advanced. • Under 'Privacy and security,' click Content settings. • Click Cookies. • Next to 'Block,' 'Clear on exit,' or 'Allow,' click Add. • Enter the web address. • To create an exception for an entire domain, insert [*.] before the domain name. For example, [*.]google.com will match drive.google.com and calendar.google.com. • You can also put an IP address or a web address that doesn't start with • Click Add. To remove an exception you don't want any more, to the right of the website, click More Remove. Note: If you’re using your Chromebook at work or school, you might not be able to change this setting. For more help,. Hello there, You recently requested an email subscription to The NICE News. We can't wait to send the updates you want via email, so please click the following link to activate your subscription immediately: (If the link above does not appear clickable or does not open a browser window when you click it, copy it and paste it into your web browser's Location bar.) As soon as your subscription is active, FeedBurner will send a daily email message if The NICE News has new content. If you did not request this subscription, or no longer wish to activate it, take no action. Simply delete this message and that will be the end of it. Cheers, The NICE News. WordPress Expert Special Offer: Free WordPress Installation, Free WordPress Development Consultation, Budget cut-off for your WordPress customization Get your own domain name for $10 only! Get the cheapest web hosting for $6 a year, Free WordPress Installation, Free WordPress Development Consultation, Budget cut-off for your WordPress customization, and much more. To test your website first. Just and we will never charge any cost, and the best part is you can also and ready to used for your website. Don’t waste your time, it’s only until end of this year! Not all visitors to your blog will be familiar with RSS feeds or how they work, while some other visitors just don’t want to mess with using a feed reader. RSS isn’t the only push-delivery method out there: email subscription is another great service to offer to your visitors. First you’ll need an email subscription service that’ll send out the emails for you. The service I recommend is. Check out my email subscription form in the sidebar to see how it looks and works. First if you don’t have one already. Then follow these steps: • Go to and login • Click “” in the upper-left-hand corner • Select your feed from the list • Click the “Publicize” tab • Click “Email Subscriptions” on the left • Make sure “Feedburner” is selected, and click “Activate” • Copy the HTML code in the box • Login to your WordPress blog’s administration area • Go to the “Appearance” section (a.k.a. “Design,” “Presentation,” or “Themes” in versions prior to 2.7) • If you’re using Widgets in your sidebar, click the “Widgets” sub-tab. Add a “Text” widget to the sidebar you want, and paste the HTML code in the configuration section. Save your changes, and that’s it! • If you’re not using widgets, you’ll need to have basic knowledge of HTML. First click the “Editor” or “Theme Editor” subpanel, and select “Sidebar” from the list. Usually sidebar modules go between tags; some other themes use. Paste the code in your sidebar between the appropriate set of tags in the location you want, click Save, and you’re done! Mar 19, 2015. The templates found in our shop come with a styled email subscription box that matches your template. This allows your readers to quickly subscribe to your blog to receive emails each time you post something new. This service is run by Feedburner and integrates with Blogger. Sometimes when you add.     OPG inhibits osteoclasts. Osteoclast Activation. Osteoclast activation stimulates bone resorption; Molecules that stimulate bone resorption. RANKL (ligand) is secreted by osteoblasts and binds to the RANK receptor on osteoclast precursor and mature osteoclast cells. PTH (secreted by many cancer cells). RANKL also evokes Ca2+ oscillations that lead to calcineurin-mediated activation of NFATc1, and therefore triggers a sustained NFATc1-dependent transcriptional program during osteoclast differentiation. We also show that NFATc1-deficient embryonic stem cells fail to differentiate into osteoclasts in response to RANKL. Background The mechanism whereby bone activates resorptive behavior in osteoclasts, the cells that resorb bone, is unknown. It is known that α v β 3 ligands are. Moved Permanently. The document has moved here. Background The mechanism whereby bone activates resorptive behavior in osteoclasts, the cells that resorb bone, is unknown. It is known that α vβ 3 ligands are important, because blockade of α vβ 3 receptor signaling inhibits bone resorption, but this might be through inhibition of adhesion or migration rather than resorption itself. Nor is it known whether α vβ 3 ligands are sufficient for resorption the consensus is that bone mineral is essential for the recognition of bone as the substrate appropriate for resorption. Methodology/Principal Findings Vitronectin- but not fibronectin-coated coverslips induced murine osteoclasts to secrete tartrate-resistant acid phosphatase, as they do on bone. Osteoclasts incubated on vitronectin, unlike fibronectin, formed podosome belts on glass coverslips, and these were modulated by resorption-regulating cytokines. Podosome belts formed on vitronectin-coated surfaces whether the substrates were rough or smooth, rigid or flexible. We developed a novel approach whereby the substrate-apposed surface of cells can be visualized in the scanning electron microscope. With this approach, supported by transmission electron microscopy, we found that osteoclasts on vitronectin-coated surfaces show ruffled borders and clear zones characteristic of resorbing osteoclasts. Ruffles were obscured by a film if cells were incubated in the cathepsin inhibitor E64, suggesting that removal of the film represents substrate-degrading behavior. Analogously, osteoclasts formed resorption-like trails on vitronectin-coated substrates. Like bone resorption, these trails were dependent upon resorbogenic cytokines and were inhibited by E64. Bone mineral induced actin rings and surface excavation only if first coated with vitronectin. Fibronectin could not substitute in any of these activities, despite enabling adhesion and cell spreading. Citation: Fuller K, Ross JL, Szewczyk KA, Moss R, Chambers TJ (2010) Bone Is Not Essential for Osteoclast Activation. PLoS ONE 5(9): e12837. Editor: Mike Klymkowsky, University of Colorado, Boulder, United States of America Received: July 22, 2010; Accepted: August 23, 2010; Published: September 17, 2010 Copyright: © 2010 Fuller et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Funding: This work was supported by St George's, University of London (). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Competing interests: The authors have declared that no competing interests exist. Introduction The osteoclast is the cell that resorbs bone. 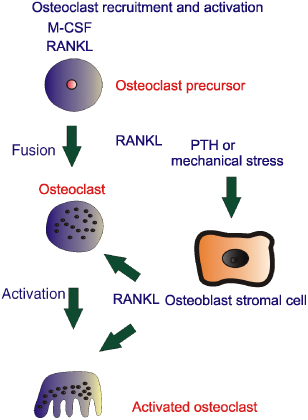
It is formed through the differentiation and fusion of mononuclear phagocyte precursors in the presence of macrophage colony-stimulating factor (M-CSF) and receptor activator of NFkB ligand (RANKL),. Its activity is normally closely integrated with that of bone-forming osteoblasts, to enable the continual removal and replacement of bone that occurs throughout life. Excessive or deficient osteoclastic function leads to a number of bone diseases, including osteoporosis and osteopetrosis. Osteoclasts resorb bone by establishing a circle of close contact between themselves and the bone surface, associated with the appearance of a ring of actin, devoid of cytoplasmic organelles (the ‘clear zone’, or ‘sealing zone’). Vesicles containing proton pumps and acid hydrolases are then inserted into the bone-apposed membrane circumscribed by this ring, throwing the membrane into convolutions: the ‘ruffled border’. Thus, a ‘resorptive hemivacuole’ is formed between cell and bone, within which protons dissolve the mineral component of bone, and acid hydrolases, predominantly cathepsin K, digest the organic matrix. Dissolved products are transported in vesicles from the resorptive hemivacuole and released at the opposite, basolateral surface,. 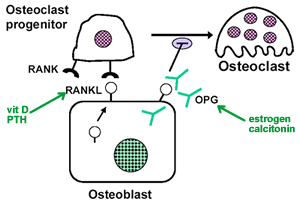
This resorptive behavior is restricted to bone. Yet it remains unknown how the osteoclast recognizes bone as appropriate for resorption. 
Ligands for the vitronectin receptor, α vβ 3 are essential, because antibodies against α vβ 3, and α vβ 3-antagonists such as echistatin and kistrin, potently inhibit bone resorption in vitro and in vivo,,,,. However, this might reflect a need for α vβ 3 ligands for attachment or migration, because these are also inhibited by α vβ 3 antagonists,. Whether α vβ 3 ligands also activate resorptive behavior and whether they are sufficient for this by themselves, has never been tested. There are alternative explanations for the induction of resorptive behavior. It might depend on some special characteristic of bone mineral: osteoclasts resorb bone if the mineral is exposed on the bone surface, but do not resorb bone that is unmineralized or has been demineralized,,. It has been proposed that activation of resorption occurs through ligation of a ‘mineral receptor’. Another suggestion is that osteoclasts are activated when they adhere to a rigid substrate, whether the adhesive ligand is vitronectin, fibronectin or collagen. Another is that it is surface roughness that is recognized. The regulation of bone resorption is normally analyzed in osteoclasts on bone. However, bone is a complex extracellular matrix, comprising not only bone mineral and collagen, but many other proteins of uncertain function. It would be very much easier to distinguish the roles of adhesive ligands, bone mineral and other factors in osteoclast activation if resorptive behavior could be assessed on a substrate other than bone. There are several correlates with resorption that could be used to enable such an analysis. One potential correlate is the induction of broad bands of F-actin, the ‘actin rings’, which are formed by osteoclasts on bone and have been shown to correlate with bone resorption,, However, osteoclasts do not form actin rings on glass/plastic substrates. Intstead they form circumferential belts of podosomes, dot-like foci of F-actin,,. There is controversy as to the relationship between podosome belts and actin rings. For example, podosomes precede actin ring formation on bone, and may be organs of attachment and migration rather than resorption; and podosome belts have a considerably greater diameter than actin rings; and consist of a discontinuous series of dots, rather than a continuous ring, of F-actin. Some have interpreted the differences between actin rings and podosome belts as indicating that the mineral component of bone is unique in its ability to induce actin rings, and is essential for resorptive behavior; others point to the strong similarities in the molecular composition of podosome belts and actin rings as evidence of their equivalence,. Although podosome belts have been noted to be suppressed by calcitonin and increased by interleukin-1 (IL-1), the effects of resorption-regulating agents on podosome belts have never been formally quantified. Thus, both the extent to which podosome belts resemble actin rings, and the extent to which possession of podosome belts signifies resorptive behavior, are uncertain. We therefore elected to further test the relationship of podosome belts to resorptive behavior in osteoclasts, before using them as markers for this activity. A second correlate is the secretion of tartrate-resistant acid phosphatase (TRAP). TRAP is highly expressed by osteoclasts, and has been shown to be secreted during bone resorption. We therefore used TRAP secretion as a marker to detect resorptive behavior on non-bone substrates. A third correlate is the formation of the ‘ruffled borders’ and ‘clear zones’ that are characteristic of resorbing osteoclasts. To detect these, we developed a novel approach whereby osteoclasts could be incubated on substrates that could be dissolved after incubation, so that the substrate-apposed surface of the osteoclast could be inspected in the scanning electron microscope (SEM). We found that podosome belt formation correlated closely with resorptive behavior in osteoclasts. We therefore used this and the other correlates of resorption to identify the characteristics of substrates that are responsible for activation of resorptive behavior in osteoclasts. We found that this activation was induced by first coating substrates with vitronectin. Vitronectin induces TRAP release by osteoclasts Osteoclasts were lifted into suspension, sedimented onto coverslips that were uncoated, or coated with fibronectin or vitronectin, and incubated for 5 hours in the resorption-inductive cytokines M-CSF, RANKL and IL-1α. After incubation, TRAP was measured in the supernatants and lysate. Because adhesion factors are required for osteoclasts to adhere to the coverslips, in particular to enable adhesion of cells in the control groups, the cells were all sedimented and subsequently incubated in 2.5% FCS. We found () that vitronectin caused a four-fold increase in TRAP release compared to controls. In contrast, fibronectin-coated coverslips suppressed TRAP release. This suppression of TRAP release by fibronectin might be because the cultures were incubated in 2.5% FCS, which contains vitronectin. Thus, while control, uncoated coverslips are available for coating by vitronectin from the FCS, this will be prevented in those coverslips previously coated with fibronectin. We repeated the TRAP secretion assay using coverslips coated with rat and human fibronectin, through the concentration range 3-50 µg/ml. TRAP secretion was similarly significantly suppressed below control levels at all concentrations (data not shown). Vitronectin induces TRAP release by osteoclasts. Bone marrow-derived osteoclasts were sedimented in MEM/2.5% FCS onto coverslips that had been coated with the vitronectin or fibronectin at the concentrations shown. After 20 minutes, the coverslips were washed and incubated for 5 hours in MEM containing 2.5% FCS, M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml), and with/without salmon calcitonin (CT) (100 pg/ml). TRAP was then measured in the supernatant and lysate. N = 5 cultures per variable. Podosome belt formation by osteoclasts is a marker for resorptive behavior Small numbers of podosome belts were observed in control cultures to which no resorption-inductive cytokines had been added (). In cultures to which osteoprotegerin (OPG), the soluble decoy receptor for RANKL, was added no podosome belts at all were observed. This suggests that the podosome belts seen in control cultures were due to residual RANKL from the preceding culture period. RANKL and IL-1α synergistically stimulated podosome belt formation. Podosome belt formation was inhibited by calcitonin (). These results are very similar to the effects of the same agents on bone resorption. Podosome belt formation is regulated in a manner that parallels regulation of bone resorption. Bone marrow-derived osteoclasts were sedimented in MEM/FCS onto glass coverslips. After 20minutes, the coverslips were washed and incubated for 5 hours in MEM/FCS in M-CSF (50 ng/ml) plus the agents shown (A), or MEM/FCS in M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml) with/without salmon calcitonin (B). A: OPG: 500 ng/ml; IL-1α: 10 ng/ml. The effect of substrate roughness on the morphology of podosome belts. Bone marrow-derived osteoclasts were sedimented in MEM/BSA onto Perspex that had been coated with vitronectin (50 µg/ml), or bone slices. Osteoclasts were incubated for 5 hours in MEM/BSA with M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml) on uncut Perspex surfaces (smooth), or on the surface of Perspex cut with the same saw as that used to prepare bone slices (rough). Phalloidin staining. Scale bars = 50 µm. Vitronectin induces podosome belt formation in osteoclasts The above results show several parallels between podosome belts and resorptive behavior. We therefore used podosome belts as a marker to test the properties of substrates that are responsible for the induction of resorptive behavior in osteoclasts. We found that significant numbers of osteoclasts adhered to vitronectin-coated glass coverslips at coating concentrations of 0.2 µg/ml and above (). Activation of these osteoclasts, as judged by the percentage of osteoclasts demonstrating podosome belts, occurred over a similar concentration range. Stimulation of podosome belts by vitronectin appeared to be due primarily to an increase in the number of belts produced. Stimulation of bone resorption shows a similar pattern: it occurs through an increase in numbers, rather than an increase in size, of excavations. Fibronectin also supported osteoclast adhesion, at similar coating concentrations. However, in contrast to the effect of vitronectin, only very small numbers of osteoclasts formed podosome belts. The number of belts showed a bimodal response, with a maximum number of belts seen at a concentration of 12.5 µg/ml. This experiment was repeated using a second batch of bovine fibronectin, and using rat and human fibronectin, with very similar results. Vitronectin induces podosome belt formation in osteoclasts. Bone marrow-derived osteoclasts were sedimented in MEM/BSA onto glass coverslips that had been coated with the vitronectin or fibronectin at the concentrations shown. The coverslips were then washed and incubated for 5 hours in MEM/BSA with M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml) before fixation and staining for F-actin and DAPI. Vitronectin (A–C) induced a dose-dependent increase in adhesion and podosome belt formation in osteoclasts. In contrast, fibronectin (D–F), effectively induced adhesion of osteoclasts, but very few of the adherent cells developed podosome belts. Vitronectin induces formation of ruffled border and clear zone in osteoclasts Osteoclasts were sedimented onto glass coverslips that had been coated with nail-varnish and then with vitronectin or fibronectin. After incubation, the cells were fixed. The disc of nail-varnish was then peeled off the coverslip, inverted onto a glass slide, dissolved in acetone, cells dehydrated in hexamethyldisilizane (HMDS), and sputter-coated for scanning electron microscopy. In preliminary experiments we tested the ability of nail-varnish to induce podosome belts. Vitronectin-coated nail-varnish induced podosome belts similar to those on vitronectin-coated glass, but no podosome belts were seen on fibronectin-coated nail-varnish (). Vitronectin induces formation of ruffled border in osteoclasts. Glass coverslips were coated with nail-varnish. This was then coated with vitronectin or fibronectin (50 µg/ml). Next, osteoclasts were incubated on this surface for 5 hours in MEM/BSA with M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml), with/without the cathepsin inhibitor E64 (3×10 −7 M) or calcitonin (100 pg/ml). A,B: Phalloidin-stained preparations after incubation on vitronectin (A) or fibronectin (B). In A, individual podosomes can be discerned within the circumferential belt of podosomes. No podosomes are seen in the osteoclast that had been incubated on fibronectin. C–H: The discs of nail-varnish were separated from the glass coverslip, inverted onto a glass slide, dissolved in acetone, dehydrated in HMDS, and sputter-coated with gold for visualization in the SEM. C,D: undersurface of osteoclast incubated on vitronectin. The central area of the undersurface of the cell is filled with dense membrane folds, while the circumference lacks folds, but shows raised foci likely to represent podosomes (arrows). Note residual film of protein attached to the cell periphery. D: higher magnification of C. Note variation in density of membrane folds in the central area. E,F: Low and higher power view of undersurface of osteoclast incubated on fibronectin. The surface is relatively featureless, and lacks the domain organization apparent after incubation on vitronectin. G: The undersurface of osteoclasts was obscured by a protein film in cultures to which the cathepsin inhibitor E64 was added. Nevertheless, a peripheral belt of raised, podosome-like structures can be discerned through the film. H: The undersurface of osteoclasts incubated with calcitonin lacked membrane ruffles and podosome belts. Scale bars: A,B: 35 µm; C,F,H: 5 µm; D,G: 2 µm; E: 20 µm. The substrate-apposed surfaces of osteoclasts incubated on vitronectin-coated substrates showed a central area of intense membrane folds surrounded by a ring of flatter membrane, upon which protrusions that appear likely to represent podosomes could be seen (). The density of the folds varied from osteoclast to osteoclast, and was generally greatest in the less spread cells. Peripheral rings of podosome-like protrusions were always seen surrounding areas that showed such folds. These appearances seem to correspond to the actin rings and ruffled borders characteristic of osteoclasts activated for resorption, and represent strong evidence that vitronectin induces resorptive behavior in osteoclasts. A full description of the morphology of the surface of the cell responsible for the resorption of bone is beyond the scope of the present work and will be submitted as a separate paper. Osteoclasts incubated on fibronectin-coated substrates were well-spread (), but in no instance were ruffles and podosome-like structures observed beneath these cells. We noted that when osteoclasts on vitronectin-coated substrates were incubated with the cathepsin inhibitor E64, the film of protein often seen without E64 was more prominent, and obscured the undersurface of the cells (). This is consistent with removal of the protein film in E64-free cultures due to the action of secreted osteoclast cathepsins. Membrane folds were not seen beneath osteoclasts incubated on vitronectin-coated substrates in the presence of calcitonin. This would be expected if the membrane folds reflect resorptive behavior, since the ruffled borders of osteoclasts disappear in the presence of calcitonin. We also inspected osteoclasts after incubation on vitronectin-coated substrates in the transmission electron microscope (TEM). We found structures characteristic of ruffled borders and clear zone in these cells (). No similar structures were seen in osteoclasts that had been incubated on fibronectin. Vitronectin induces ruffled borders and clear zones in osteoclasts. Osteoclasts were incubated for 5 hours in MEM/BSA with M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml) in 6-well plate wells coated with vitronectin or fibronectin (50 µg/ml), before raising into suspension with a cell scraper and preparation for TEM. A: Osteoclast incubated on vitronectin shows a central area of ruffled border (arrowhead) and a peripheral area free of organelles (‘clear zone’) (arrows). B, C: higher magnification of center (B) and lower portion (C) respectively of A, showing area of ruffled border (B) and clear zone (C); D–F: Osteoclast incubated on fibronectin shows well-spread appearance, but the undersurface lacked the membrane folds and clear zones seen in osteoclasts incubated on vitronectin. E and F are from central and lower portion of D respectively. Scale bars: A, D: 5 µm; B, C, E, F: 1 µm. Vitronectin induces formation of resorption-like trails by osteoclasts on glass substrates We analyzed further the hypothesis that digestion of the protein film noted above reflects resorptive behavior. For this, osteoclasts were sedimented onto vitronectin- or fibronectin-coated glass coverslips, incubated with/without resorption-inducing cytokines and protease inhibitors and examined in the SEM. We found that many of osteoclasts incubated in resorption-stimulating cytokines showed sharp-edged cleared areas behind and below their retreating margins (). Such areas were not seen in association with the macrophagic cells (smaller cells, with leaf-like rather than filopodial dorsal membrane folds). These sharp-edged cleared areas were reminiscent of the excavations seen behind the retreating margin of osteoclasts incubated on bone. In contrast, cleared areas were rare in cultures from which RANKL and IL-1α had been omitted, and those that were seen were substantially smaller (). Formation of cleared areas was inhibited by the cathepsin-inhibitor E64, but the metalloproteinase inhibitor GM6001 had no apparent effect. Vitronectin induces formation of resorption-like trails by osteoclasts on glass substrates. Osteoclasts were incubated for 5 hours in MEM/BSA with M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml) (A, C–F), or M-CSF (B), together with the cathepsin inhibitor E64 (3×10 −7 M) (C) or GM6001 (1.3×10 −5 M) (D), on glass coverslips coated with vitronectin (A–D) or fibronectin (E, F) (50 µg/ml), before preparation for SEM. A: Osteoclasts incubated in resorption-inducing cytokines on vitronectin show well-defined cleared areas at the retreating margins of the cells. Macrophagic cells (some of which are identified by arrows, as the smaller cells, with leaf-like, rather than filopodial membrane folds) are not associated with cleared areas. B: Only occasional, and small, cleared areas were seen in cultures to which resorption-inducing cytokines had not been added. C: The formation of cleared areas was inhibited by the cysteine protease inhibitor E64. D: the MMP inhibitor GM6001 was without apparent effect. E, F: No cleared areas were seen at the retreating pole of osteoclasts incubated on fibronectin, although there was evidence of focal disturbance to the surface of the protein film in the region of filopodia. Scale bars = 50 µm. No cleared resorption-like trails were seen in association with the retreating margin of osteoclasts incubated on fibronectin, although the protein film had sometimes been removed in small discontinuous foci adjacent to filopodia (). Vitronectin coating of anorganic bone enables resorptive behavior in osteoclasts We found that osteoclasts did not adhere to anorganic bone that had not been coated with adhesion factor. After coating with vitronectin, many osteoclasts had adhered and many of these showed podosome belts (). Excavations could be seen in the SEM after removal of cells. Osteoclasts adhered to fibronectin-coated anorganic bone in similar numbers, and spread well, but neither podosome belts nor excavations were formed by any of the cells (). Vitronectin coating of anorganic bone enables podosome belt formation and resorption. Osteoclasts were incubated for 5 hours in MEM/BSA with M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml) on slices of anorganic bone coated with vitronectin or fibronectin (50 µg/ml). Podosome belts and excavations were frequently seen on vitronectin-coated anorganic bone slices (A, C), but were never seen on fibronectin-coated anorganic bone slices (B, D). A, B: Phalloidin/DAPI staining; C, D: SEM images. Scale bars A–C: 25 µm; D: 50 µm. We attempted to assess the undersurface of cells incubated on slices of vitronectin-coated anorganic bone, by dissolving the bone mineral in EDTA after fixation. However, the vitronectin had percolated through the specimen, so that a three-dimensional proteinaceous sponge-like cast of the anorganic bone slice remained beneath the cells. Flexible substrates can induce podosome belt formation To test whether podosome belt formation requires a rigid substrate, vitronectin was conjugated or coated onto PDMS sheets or silicone rubber films respectively. We found that vitronectin strongly induced podosome belts in both of these flexible substrates (). Explanations for the differing size of podosome rings on these two substrates might include differences in surface roughness, which has been shown to influence the size of actin rings, or differences in the density of vitronectin bound by the two substrates. Flexible substrates can induce podosome belt formation. Osteoclasts were incubated for 5 hours in MEM/BSA with M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml) on PDMS sheets or silicone films coated with vitronectin or fibronectin (50 µg/ml). A,B: Osteoclasts adhered to fibronectin-coated PDMS sheets in large numbers, but podosome belts were very rare. In contrast, a similar proportion of osteoclasts formed podosome belts on vitronectin-coated PDMS sheets to our previous experience using rigid substrates. Discussion Although much has been learned over the last decade about osteoclastic differentiation, far less is known of the mechanisms through which bone induces resorption. Vitronectin receptor ligands have been shown to be necessary for bone resorption,,,,, but it is not known whether this reflects a requirement for adhesion, cell spreading, migration and/or activation of bone resorption. Nor is it known whether these ligands are sufficient to activate resorption by osteoclasts. There are several alternative possibilities. Activation of resorption might depend on some special characteristic of bone mineral: osteoclasts resorb bone if the mineral is exposed; and it has been reported that actin rings, which correlate with resorptive behavior, are formed by osteoclasts only on bone or bone mineral. An osteoclastic ‘mineral receptor’ has been proposed. Another suggestion is that osteoclasts are activated when they detect a rigid substrate, whether the adhesive ligand is vitronectin or fibronectin. Another is that rough surfaces stimulate resorption. To analyze the mechanism of activation of resorptive behavior in osteoclasts, our approach has been to incubate osteoclasts on substrates other than bone, and to test the characteristics of the substrate that lead to behavior expected of or observed in resorbing osteoclasts. For these experiments, we exploited a technique we have recently developed whereby osteoclasts can be lifted into suspension and can excavate bone within 3 hours of sedimentation. This enables us to test the response of mature osteoclasts to defined substrates that have not been conditioned by the medium used to generate osteoclasts, or by the osteoclasts themselves. We used these suspensions first to test the ability of substrates to induce secretion of TRAP. We chose this approach because TRAP is released into the resorptive hemivacuole during bone resorption, whence it is endocytosed and then released at the basolateral surface of the cell. TRAP secretion by osteoclasts sedimented onto bone slices can be detected in the supernatant and correlates with bone resorption. Therefore, if substrates other than bone are capable of inducing resorptive behavior, TRAP secretion should occur. We observed potent stimulation of TRAP release by osteoclasts incubated on vitronectin-coated glass coverslips. This was abolished by the resorption-inhibiting hormone calcitonin. These results are consistent with the hypothesis that vitronectin activates resorptive behavior. In contrast, fibronectin not only failed to induce, but actually suppressed TRAP secretion. Small quantities of serum were used in the assay, to enable adhesion of osteoclasts in the control group. Because serum contains vitronectin, the experiment is in reality comparing lower with higher vitronectin densities. From this perspective, the explanation for inhibition of TRAP secretion by fibronectin might be that fibronectin prevents vitronectin in the serum from binding to the vitronectin receptor. Alternatively, the explanation might be that serum-derived vitronectin cannot coat substrates already coated with fibronectin. We noted a similar propensity of higher concentrations of fibronectin to suppress ‘basal’ podosome belt formation, in cultures to which no serum had been added (). The explanation for this might be that osteoclast-derived osteopontin, a potent α vβ 3 ligand, might be excluded from the substrate by fibronectin. A further intriguing possible explanation for the results of both experiments is that suppression of basal osteoclast activation is caused by the mutual antagonism of signaling mechanisms demonstrated by integrins, such that dominant signaling by higher densities of fibronectin will suppress activation of the vitronectin receptor. Whatever the explanation is, the differing ability of these ligands to activate osteoclasts, despite very similar actions as adhesion factors, is striking. During bone resorption osteoclasts form a circumferential, actin-rich ‘sealing zone’ or ‘clear zone’. This ‘actin ring’ has been correlated with bone resorption,. Osteoclasts on non-mineralized substrates do not show actin rings, but rather circumferential belts of podosomes. The extent to which podosome belts resemble actin rings and to which possession of podosome belts signifies resorptive behavior, is uncertain. It has been claimed that the sealing zone on bone has a different three- dimensional organization that is not derived from podosomes,. However, it has also been suggested that isolated podosomes fuse to give rise to a continuous sealing zone,. This view is supported by recent evidence that the sealing zone consists of structural units clearly related to individual podosomes, which differ primarily in density and inter-connectivity from the podosome belts observed on other substrates. We have identified two further similarities between podosome belts and actin rings. First, podosome belts seem more closely related to bone resorption than to cell spreading and migration. Thus, the effects of regulators on podosome belts closely paralleled their known effects on bone resorption. In contrast, neither M-CSF, which induces migration and spreading but inhibits bone resorption, nor fibronectin, which facilitated adhesion and spreading, increased podosome belt formation. Second, we have found a possible explanation for a notable difference between podosome belts and actin rings, namely their circumference. Typically, the spreading of cells such as fibroblasts and epithelial cells is greater on smooth than on rough surfaces,. To see whether the difference in diameter of podosome belts could be attributed to surface roughness, we compared podosome belts formed on a slice of Perspex cut with our bone saw, with those formed on a smooth, uncut surface from the same block. We found that the belts were of substantially smaller diameter on the sawn surface, and more like actin rings on bone. Thus, the lower density of podosome subunits on glass might reflect the greater spreading caused by the smooth surface. Increased cell spreading would be expected to simultaneously increase podosome belt diameter and decrease podosome density. It has recently been shown that greater roughness in calcite surfaces stabilizes and enlarges actin structures in osteoclasts. This implies that surface roughness may play a role in the regulation of resorption by osteoclasts. To the extent that we found that rough surfaces made actin structures more closely resemble those seen on bone, our data also support the notion that resorption is enhanced on rough surfaces. However, we have also presented strong evidence that activation of resorptive behavior can occur on smooth, vitronectin-coated surfaces. Therefore, a surface does not necessarily have to be rough to activate osteoclasts. In contrast to the observations of Geblinger et al, discussed above, we found that a smooth surface enlarged actin structures. There might be a bimodal relationship between actin structure diameter and surface roughness: spreading by osteoblasts has been found to be reduced both above and below an ‘optimal’ roughness value. In any event, our data show that the differing diameters of actin structures on glass and bone do not necessarily imply that podosome belts and actin rings are functionally distinct structures. This supports the notion that podosome belts reflect activation of resorptive behavior in osteoclasts. We found that vitronectin induced podosome belt formation, while fibronectin did not. While there have been several reports documenting the ability of a variety of adhesion molecules to enable osteoclastic adhesion to substrates, there has been only one previous comparison of the ability of adhesion molecules to activate actin ring formation. In this report, it was found that fibronectin induces podosome belts, but in our experiments neither rat, bovine nor human fibronectin did so, despite good bioactivity as an adhesion and spreading factor. The cells used in the previous report were mixed populations derived from co-culture with osteoblasts, and used a substantially longer incubation period. These differences would predispose to secretion of vitronectin receptor ligands onto the glass surface, by osteoblastic cells or by the osteoclasts themselves. The contrast between the effects of vitronectin and fibronectin on osteoclasts is remarkable. The osteoclast expresses both the vitronectin receptor α vβ 3 and the fibronectin receptor α 5β 1, and both vitronectin and fibronectin enabled osteoclasts to attach to glass coverslips or bone mineral in the absence of serum, and both facilitated cell spreading. Yet only vitronectin was able to induce resorptive behavior. It has been established that bone resorption is dependent upon c-Src,,, so that the likely molecular basis for our observations is the demonstration that c-Src binds selectively to β 3 not β 1 integrins, and that clustering of β 3 in vivo activates c-Src, while c-Src deficiency has no detectable effects on fibronectin-receptor function. During bone resorption, osteoclasts show a characteristic ‘ruffled border’, consisting of membrane folds, circumscribed by a ‘clear zone’. Our TEM and SEM studies showed strong morphological evidence, for the first time, of the formation of ruffled borders and clear zones on a non-mineral substrate. Like podosome belts and release of TRAP, these characteristic structures were induced by vitronectin but not by fibronectin. We noted in the SEM that substrates were covered by a protein film. This film was more apparent under osteoclasts incubated in the cathepsin inhibitor E64. Similar degradation of extracellular matrix by osteoclasts has previously been reported. This matrix degradation might be a consequence of cell spreading or migration, or it might reflect resorptive behavior. Our data favor the latter: the film was digested by osteoclasts on vitronectin-coated, but not fibronectin-coated glass coverslips, despite similar cell spreading; and like bone resorption, digestion was less pronounced in M-CSF (which stimulates osteoclast motility and spreading but inhibits bone resorption ), than in resorption-stimulators. The effects of proteinase inhibitors also suggest that the trails are a consequence of resorptive behavior rather than motility: degradation was inhibited by the cathepsin inhibitor but not by the metalloproteinase inhibitor. These observations are consistent with the hypothesis that vitronectin-coated substrates induce resorptive behavior in osteoclasts. The consensus has been that bone mineral is essential for resorptive behavior in osteoclasts. Thus, if the mineral component of bone is removed, neither bone resorption nor actin rings are observed,. In contrast, osteoclasts resorb bone from which the organic component has been removed (bone mineral), and some other mineral substrates,,. These observations have led to the suggestion that osteoclasts recognize bone through a putative ‘mineral receptor’. It should be noted though that previous experiments included serum in the incubation medium. In the present experiments we used serum-free medium. We found without serum osteoclasts did not adhere to bone mineral, and while both vitronectin and fibronectin enabled adhesion, only vitronectin induced podosome belts and substrate resorption. Thus, vitronectin can act at least as a cofactor (with bone mineral) for induction of resorption by osteoclasts. However, we also found that coating non-mineral substrates with vitronectin induces TRAP release, podosome belt formation, protein coat degradation and ruffled border formation. This shows that it is the vitronectin, rather than the bone mineral, that is necessary for induction of resorptive behavior. Bone mineral appears to act, like glass coverslips, through its ability to immobilize vitronectin receptor ligands. Vitronectin receptor ligands such as osteopontin and thrombospondin are highly expressed by bone cells and have a strong affinity for bone mineral through a multiphosphorylated motif,,,. Such ligands, incorporated in bone during bone formation, might activate osteoclasts when bone mineral is exposed by osteoblastic cells. Alternatively or additionally, activation might follow binding of osteopontin or bone sialoprotein, which are known to be expressed by osteoclasts,,,, to freshly-exposed bone mineral, or binding of α vβ 3 ligands secreted by osteoclast-regulatory osteoblastic cells independently of bone formation, or vitronectin from serum, in which it is present at between 300–700 µg/ml,. Osteoblastic cells are known to be crucial for the regulation of osteoclast formation and the rate of their resorptive activity. The above observations provide a novel mechanism through which osteoblastic cells could extend their ability to regulate osteoclasts to include the induction and localization of resorption, by exposing mineral onto the bone surface through secretion of interstitial collagenase. Glass and bone share not only the ability to adsorb vitronectin, but structural rigidity. It has been suggested that structural rigidity is what distinguishes tissues that are resorbed from those that are not. This might explain why demineralized bone does not induce bone resorption or podosome belts,. Our observations do not support this model. We found that, unlike vitronectin, fibronectin coating of rigid substrates does not induce resorptive behavior despite a similar capacity to induce adhesion and spreading. This suggests that high substrate rigidity is not sufficient to induce resorptive behavior. Further, we noted that flexible silicone and PDMS sheets induced podosome belts in osteoclasts, when coated with vitronectin. Thus, a rigid substrate is neither necessary nor sufficient for the induction of resorptive behavior. In fact, vitronectin appears to be capable of recapitulation of the osteoclast-bone interaction: it is sufficient to induce osteoclasts to form podosome belts, to secrete enzymes, to undertake substrate digestion, and to form clear zones and ruffled borders. Vitronectin receptor ligands have long been known to be necessary for bone resorption, but the finding that they are sufficient is novel. Moreover, it has not been previously determined whether vitronectin is needed for activation of resorption, and/or for other functions essential for bone resorption to occur, such as cell adhesion. It has been shown in many elegant molecular biological studies that the vitronectin receptor participates in cell signaling in osteoclasts,,. Our experiments should facilitate such studies by enabling a distinction between those signals that mediate adhesion and cell spreading and those that mediate the induction of bone resorption. We have found a strong correlation between podosome belts and other correlates of resorptive behavior, and this makes them a reliable indicator of resorptive activity in osteoclasts. We also show that α vβ 3 ligands are not only necessary but sufficient for the induction of resorptive behavior in osteoclasts; and that it is these ligands, rather than bone itself, that induces resorption; and that bone is recognized due to the high affinity of bone mineral for these ligands combined with the specific signals generated by ligation of α vβ 3, rather than by its mechanical or topographical attributes or through a putative ‘mineral receptor’. Last, we present a novel approach whereby the substrate-apposed surface of not only osteoclasts but any substrate-adherent cell can be directly inspected in the SEM. Media and reagents Cells were incubated in minimum essential medium (MEM) with Earle's salts, supplemented with 10% fetal calf serum (FCS), 2 mM glutamine, 100 IU/ml benzylpenicillin and 100 µg/ml streptomycin (all Sigma, Poole, Dorset, UK) for osteoclast formation. For assays, 10% FCS was replaced with 2.5% FCS for TRAP release, or 0.1% BSA (Sigma) for all other experiments, unless stated otherwise. Recombinant human M-CSF, recombinant human OPG and recombinant mouse RANKL were purchased from PeproTech EC (London, UK). Recombinant mouse IL-1α and purified human TGF-β1 were obtained from R & D Systems (Abingdon, Oxon., UK). GM6001 was from Merck Chemicals Ltd. (Nottingham, UK). All other reagents were provided by Sigma, unless otherwise stated. Incubations were performed at 37°C in 5% CO 2 in humidified air, unless stated otherwise. Preparation of vitronectin- and fibronectin-coated substrates Slices of bovine cortical bone were prepared as previously described. These were rendered anorganic by treating with sodium hypochlorite solution (10–15%) for 8 days, followed by extensive washing in water. Perspex slices (ca. 1 cm 2) were prepared from a block of Perspex by cutting slices in the same low-speed saw as was used to prepare bone slices. Slices cut from the uncut surface of the block were used as smooth substrates and compared with the cut surface of deeper slices. Thirteen mm diameter glass coverslips were coated with a film of clear nail-varnish (Rimmel, London, UK) using a Pasteur pipette. Polydimethylsiloxane (PDMS) rubber sheets were prepared by casting ELASTOSIL RT 601 (Wacker Chemie AG, Munich, Germany). The two components were mixed according to the manufacturer's instructions and 0.5 ml added to wells of 12-well plates (BD Biosciences, Oxford, UK), before being de-gassed using a vacuum pump for 30 minutes. The silicone rubber was allowed to cure for 3 days at room temperature. Flexible silicone rubber films were prepared by adding 25 µl silicon DC-200 to the centre of 6-well plate wells (Greiner Bio-One, Stonehouse, Gloucestershire, UK) and allowing to spread for 18 h. The surface of the silicone was then polymerized by sputter coating with gold (20 mA; 20 seconds). To coat the substrates, vitronectin or fibronectin (both bovine unless stated otherwise) were dissolved in water. Fifty µl was placed on the surface of 6 mm diameter coverslips or the centre of the silicone films, and 300 µl on 13 mm diameter glass coverslips, Perspex or anorganic bone slices (ca. 1 cm 2), and dried overnight at room temperature in a tissue culture hood. Vitronectin or fibronectin were covalently bound to the surface of PDMS sheets using sulfo-SANPAH (Thermo Scientific, Basingstoke, Hampshire, UK). Briefly, 1 ml of 1 mM sulfo-SANPAH was added to wells containing the sheets and bound to the surface of the silicone rubber by exposure to UV light (365 nM, 15 minutes) in a UV cross-linker (model CL-E508; UVITEC, Cambridge, UK). The sulfo-SANPAH was then removed, the PDMS sheets washed with PBS, fresh sulfo-SANPAH added and the photoactivation process repeated. After extensive washing, 1.5 ml of vitronectin or fibronectin (both 30 µg/ml PBS) were added onto the PDMS sheets and the proteins allowed to bind for 18 hours at room temperature. The sheets were washed with PBS and 1.5 ml MEM/BSA added to wells prior to cell addition. Preparation of osteoclast suspensions MF1 mice (4–8 weeks old) were killed by cervical dislocation. Femora and tibiae were aseptically removed and dissected free of adherent soft tissue. The bone ends were removed and the marrow cavity flushed out into a petri dish by slowly injecting PBS at one end of the bone using a sterile 25-gauge needle. The bone marrow suspension was passed repeatedly through a 21-gauge needle to obtain a single cell suspension. Bone marrow cells were then washed, resuspended in MEM/FCS and incubated at a density of 3×10 5 cells/ml for 24 hours in a 175-cm 2 flask (Greiner Bio-One) with M-CSF (5 ng/ml), to deplete the cell preparations of stromal cells. Non-adherent cells were collected by centrifugation and added to 90 mm diameter cell culture dishes (Greiner Bio-One) in MEM/FCS, containing M-CSF (50 ng/ml), RANKL (30 ng/ml) and TGF-β (0.1 ng/ml) (7.2×10 6 cells in 25 ml for each dish). Cultures were incubated for 5 days, by which time osteoclast formation was maximal. Cells were fed every 2–3 days by replacing 15 ml of culture medium with an equal volume of fresh medium and cytokines. After formation of osteoclasts on the base of a 90 mm-diameter plastic tissue culture dish, osteoclasts were scraped from the dish into suspension as previously described. To do this, the medium was removed and the cell layer washed 3 times with PBS without calcium and magnesium. Six ml of 0.02% EDTA was added to the dish and cells incubated for 20 minutes at room temperature. The EDTA was then removed from the dish and replaced with 3.6 ml of calcium/magnesium-free PBS. A cell scraper (Greiner Bio-One) was used to scrape the cells into the PBS, and the resulting cell suspension was agitated using a pipette to ensure uniform cell dispersal, and added to cultures as described below. Measurement of release of TRAP Vitronectin- or fibronectin-coated or uncoated 6 mm diameter coverslips were placed in the wells of a 96-well plate (Greiner Bio-One) and 75 µl MEM/2.5% FCS was added to each well. Seventy five µl of the osteoclast suspension was then added to each well. Cells were allowed sediment for 20 minutes at 37°C before the coverslips were washed and transferred to fresh 96-well plate wells and incubated for 5 hours in 100 µl MEM/2.5% FCS in M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml). After incubation of cells for 5 hours on coverslips, supernatants were removed for measurement of enzyme release. Cell lysates, for assessment of enzyme remaining in cells, were then prepared: coverslips were washed in PBS, transferred to fresh 96-well plate wells and incubated in 100 µl 0.1% Triton X-100 in water (v/v) for 10 minutes. TRAP enzyme activity was measured by the conversion of p-nitrophenyl phosphate to p-nitrophenol in the presence of sodium tartrate. Eighty µl of each supernatant or lysate, diluted appropriately, was added to 96-well plate wells containing 80 µl 0.09 M citrate buffer with 20 mM phosphatase substrate and 80 mM tartaric acid and incubated at room temperature for 40 minutes. The reaction was stopped by addition of 40 µl of 0.5 M sodium hydroxide. Optical absorbance was measured at 405 nm on an Opsys MR plate reader (Thermo Scientific) against a standard curve of p-nitrophenol. The extent of enzyme activity released into the supernatant as a percentage of total enzyme activity in the supernatant and lysate combined was calculated for each culture assayed. Assessment of podosome belts in osteoclasts Vitronectin- or fibronectin-coated or uncoated bone, 13 mm diameter glass and Perspex substrates were placed in the wells of a 24-well-plate (Greiner Bio-One) containing 450 µl MEM/BSA or MEM/FCS. Four hundred and fifty µl of the osteoclast suspension was added to each well. Cells were allowed sediment for 20 min at 37°C before the substrates were washed and transferred to fresh 24-well plate wells and incubated for 5 h in 1 ml MEM/BSA or MEM/FCS with M-CSF (50 ng/ml), with/without RANKL (30 or 100 ng/ml), IL-1α (10 ng/ml), OPG (500 ng/ml) and salmon calcitonin (0.1–1000 pg/ml). Two or 1.5 ml MEM/BSA were added to wells containing silicone rubber films or PDMS sheets respectively and an equal volume of osteoclast cell suspension added. Cells were allowed to sediment for 20 minutes then substrates washed 3 times with PBS. Cells were incubated 5 hours in 2 ml (films) or 1.5 ml (sheets) MEM/BSA with M-CSF (50 ng/ml), RANKL (30 ng/ml) and IL-1α (10 ng/ml). After incubation for 5 hours on these substrates, the cultures were fixed in 10% formalin and cells permeabilized with 0.1% Triton X-100 for 5 minutes. Cells were then incubated in 1 µg/ml FITC-conjugated phalloidin for 45 minutes at 37°C, washed and mounted in VECTORSHIELD mounting medium with DAPI (Vector Laboratories, Peterborough, UK). Podosome belts were counted by photographing a minimum of 10 fields/replicate using a x10 objective on a Zeiss Axiovert 200 M microscope (Carl Zeiss, Welwyn Garden City, Hertfordshire, UK) using a Zeiss Axiocam MRC5 camera and Zeiss Axiovision 4.6 software. The number of osteoclasts, the percentage of osteoclasts with podosome belts and the total area within podosome belts were quantified for each photograph. Assessment of osteoclast morphology in the SEM Nail-varnish coated coverslips that had been coated with vitronectin or fibronectin were placed in 24-well plates. Osteoclast suspension was added as above, and incubated for 5 hours. The coverslips were then washed, and fixed in 4% glutaraldehyde in 0.2 M sodium cacodylate buffer for 18 hours. After fixation the circle of nail-varnish could readily be detached from the glass coverslip, and it was inverted onto a glass microscope slide. The nail-varnish was dissolved and the cells dehydrated in an acetone series (30, 50, 70, 90, 100%; 5 min each). Care was taken to avoid allowing the circle dry out. After 5 minutes in 100% acetone, HMDS was added gently to the cells, and after a further 5 minutes the slide was drained and allowed to dry. The glass slide was cut to a square, attached to a stub, sputter-coated with gold and examined in a Cambridge Stereoscan 360 SEM (Cambridge Instrument Company, Cambridge, UK). Assessment of osteoclast morphology in the TEM Six-well plate wells were coated with vitronectin or fibronectin (both 1.5 ml/well at 30 µg/ml) and 2 ml MEM/BSA added to wells after coating prior to cell addition. Two ml of osteoclast cell suspension was then added to the wells and cells sedimented and incubated as above. After incubation, cells were fixed in 4% glutaraldehyde in 0.2 M cacodylate buffer for 18 hours. The cell layers were then washed 3 times with PBS, scraped into 1 ml of PBS and then processed for TEM. Assessment of resorption-like trails by osteoclasts Vitronectin- or fibronectin-coated glass coverslips were placed in 24-well plate wells with 450 µl MEM/BSA and 450 µl cell suspension, diluted 1∶4 with PBS, added as above. After cell sedimentation, the coverslips were incubated 5 hours in 1 ml MEM/BSA with M-CSF (50 ng/ml) with/without RANKL (30 ng/ml), IL-1α (10 ng/ml), E64 (3×10 −7 M) and GM6001 (13 µM). After incubation, the coverslips were washed in PBS and fixed in 4% glutaraldehyde 18 hours. Coverslips were then washed in PBS and cells prepared for SEM by dehydrating through an acetone series and HMDS, and sputter coating as above. References • 1. Chambers TJ (2000) Regulation of the differentiation and function of osteoclasts. J Pathol 192: 4–13.TJ Chambers2000Regulation of the differentiation and function of osteoclasts.J Pathol192413 • • • • 2. Suda T, Takahashi N, Udagawa N, Jimi E, Gillespie MT, et al. (1999) Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr Rev 20: 345–357.T. JimiMT Gillespie1999Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families.Endocr Rev20345357 • • • • 3. Vaananen HK, Zhao H, Mulari M, Halleen JM (2000) The cell biology of osteoclast function. J Cell Sci 113(Pt 3): 377–381.HK VaananenH. MulariJM Halleen2000The cell biology of osteoclast function.J Cell Sci113Pt 3377381 • • • • 4. Stenbeck G (2002) Formation and function of the ruffled border in osteoclasts. Semin Cell Dev Biol 13: 285–292.G. Stenbeck2002Formation and function of the ruffled border in osteoclasts.Semin Cell Dev Biol13285292 • • • • 5. Fisher JE, Caulfield MP, Sato M, Quartuccio HA, Gould RJ, et al. (1993) Inhibition of Osteoclastic Bone-Resorption Invivo by Echistatin, an Arginyl-Glycyl-Aspartyl (Rgd)-Containing Protein. Endocrinology 132: 1411–1413.JE FisherMP CaulfieldM. SatoHA QuartuccioRJ Gould1993Inhibition of Osteoclastic Bone-Resorption Invivo by Echistatin, an Arginyl-Glycyl-Aspartyl (Rgd)-Containing Protein.Endocrinology3 • • • • 6. Lakkakorpi PT, Horton MA, Helfrich MH, Karhukorpi EK, Vaananen HK (1991) Vitronectin Receptor Has a Role in Bone-Resorption but Does Not Mediate Tight Sealing Zone Attachment of Osteoclasts to the Bone Surface. Journal of Cell Biology 115: 1179–1186.PT LakkakorpiMA HortonMH HelfrichEK KarhukorpiHK Vaananen1991Vitronectin Receptor Has a Role in Bone-Resorption but Does Not Mediate Tight Sealing Zone Attachment of Osteoclasts to the Bone Surface.Journal of Cell Biology6 • • • • 7. Engelman VW, Nickols GA, Ross FP, Horton MA, Griggs DW, et al. (1997) A peptidomimetic antagonist of the alpha(v)beta(3) integrin inhibits bone resorption in vitro and prevents osteoporosis in vivo. Journal of Clinical Investigation 99: 2284–2292.VW EngelmanGA NickolsFP RossMA HortonDW Griggs1997A peptidomimetic antagonist of the alpha(v)beta(3) integrin inhibits bone resorption in vitro and prevents osteoporosis in vivo.Journal of Clinical Investigation • • • • 8. Chambers TJ, Fuller K, Darby JA, Pringle JA, Horton MA (1986) Monoclonal antibodies against osteoclasts inhibit bone resorption in vitro. Bone Miner 1: 127–135.TJ ChambersK. FullerJA DarbyJA PringleMA Horton1986Monoclonal antibodies against osteoclasts inhibit bone resorption in vitro.Bone Miner1127135 • • • • 9. Horton MA, Taylor ML, Arnett TR, Helfrich MH (1991) Arg-Gly-Asp (RGD) peptides and the anti-vitronectin receptor antibody 23C6 inhibit dentine resorption and cell spreading by osteoclasts. Exp Cell Res 195: 368–375.MA HortonML TaylorTR ArnettMH Helfrich1991Arg-Gly-Asp (RGD) peptides and the anti-vitronectin receptor antibody 23C6 inhibit dentine resorption and cell spreading by osteoclasts.Exp Cell Res195368375 • • • • 10. Nakamura I, Pilkington MF, Lakkakorpi PT, Lipfert L, Sims SM, et al. (1999) Role of alpha(v)beta(3) integrin in osteoclast migration and formation of the sealing zone. J Cell Sci 112(Pt 22): 3985–3993.I. NakamuraMF PilkingtonPT LakkakorpiL. LipfertSM Sims1999Role of alpha(v)beta(3) integrin in osteoclast migration and formation of the sealing zone.J Cell Sci112Pt • • • • 11. Holliday LS, Welgus HG, Fliszar CJ, Veith GM, Jeffrey JJ, et al. (1997) Initiation of osteoclast bone resorption by interstitial collagenase. J Biol Chem 272: 8.LS HollidayHG WelgusCJ FliszarGM VeithJJ Jeffrey1997Initiation of osteoclast bone resorption by interstitial collagenase.J Biol Chem058 • • • • 12. Chambers TJ, Thomson BM, Fuller K (1984) Effect of substrate composition on bone resorption by rabbit osteoclasts. J Cell Sci 70: 61–71.TJ ChambersBM ThomsonK. Fuller1984Effect of substrate composition on bone resorption by rabbit osteoclasts.J Cell Sci706171 • • • • 13. Chambers TJ, Fuller K (1985) Bone cells predispose bone surfaces to resorption by exposure of mineral to osteoclastic contact. J Cell Sci 76: 155–165.TJ ChambersK. Fuller1985Bone cells predispose bone surfaces to resorption by exposure of mineral to osteoclastic contact.J Cell Sci76155165 • • • • 14. Saltel F, Destaing O, Bard F, Eichert D, Jurdic P (2004) Apatite-mediated actin dynamics in resorbing osteoclasts. Mol Biol Cell 15: 5231–5241.F. Jurdic2004Apatite-mediated actin dynamics in resorbing osteoclasts.Mol Biol Cell • • • • 15. Nakamura I, Takahashi N, Sasaki T, Jimi E, Kurokawa T, et al. (1996) Chemical and physical properties of the extracellular matrix are required for the actin ring formation in osteoclasts. J Bone Miner Res 11: 1873–1879.I. Kurokawa1996Chemical and physical properties of the extracellular matrix are required for the actin ring formation in osteoclasts.J Bone Miner Res • • • • 16. Geblinger D, Addadi L, Geiger B (2010) Nano-topograpy sensing by osteoclasts. Geiger2010Nano-topograpy sensing by osteoclasts.J Cell Sci • • • • 17. Lakkakorpi PT, Vaananen HK (1990) Calcitonin, prostaglandin E2, and dibutyryl cyclic adenosine 3′,5′-monophosphate disperse the specific microfilament structure in resorbing osteoclasts. J Histochem Cytochem 38: 1487–1493.PT LakkakorpiHK Vaananen1990Calcitonin, prostaglandin E2, and dibutyryl cyclic adenosine 3′,5′-monophosphate disperse the specific microfilament structure in resorbing osteoclasts.J Histochem Cytochem • • • • 18. Burgess TL, Qian Y, Kaufman S, Ring BD, Van G, et al. (1999) The ligand for osteoprotegerin (OPGL) directly activates mature osteoclasts. J Cell Biol 145: 527–538.TL BurgessY. KaufmanBD RingG. Van1999The ligand for osteoprotegerin (OPGL) directly activates mature osteoclasts.J Cell Biol145527538 • • • • 19. Lakkakorpi PT, Vaananen HK (1996) Cytoskeletal changes in osteoclasts during the resorption cycle. Microsc Res Tech 33: 171–181.PT LakkakorpiHK Vaananen1996Cytoskeletal changes in osteoclasts during the resorption cycle.Microsc Res Tech33171181 • • • • 20. Turksen K, Kanehisa J, Opas M, Heersche JN, Aubin JE (1988) Adhesion patterns and cytoskeleton of rabbit osteoclasts on bone slices and glass. J Bone Miner Res 3: 389–400.K. OpasJN HeerscheJE Aubin1988Adhesion patterns and cytoskeleton of rabbit osteoclasts on bone slices and glass.J Bone Miner Res3389400 • • • • 21. Teti A, Marchisio PC, Zallone AZ (1991) Clear zone in osteoclast function: role of podosomes in regulation of bone-resorbing activity. Am J Physiol 261: C1–7.A. TetiPC MarchisioAZ Zallone1991Clear zone in osteoclast function: role of podosomes in regulation of bone-resorbing activity.Am J Physiol261C17 • • • • 22. Jurdic P, Saltel F, Chabadel A, Destaing O (2006) Podosome and sealing zone: specificity of the osteoclast model. Eur J Cell Biol 85: 195–202.P. Destaing2006Podosome and sealing zone: specificity of the osteoclast model.Eur J Cell Biol85195202 • • • • 23. Saltel F, Chabadel A, Bonnelye E, Jurdic P (2008) Actin cytoskeletal organisation in osteoclasts: a model to decipher transmigration and matrix degradation. Eur J Cell Biol 87: 459–468.F. Jurdic2008Actin cytoskeletal organisation in osteoclasts: a model to decipher transmigration and matrix degradation.Eur J Cell Biol87459468 • • • • 24. Luxenburg C, Geblinger D, Klein E, Anderson K, Hanein D, et al. (2007) The architecture of the adhesive apparatus of cultured osteoclasts: from podosome formation to sealing zone assembly. PLoS One 2: e179.C. Hanein2007The architecture of the adhesive apparatus of cultured osteoclasts: from podosome formation to sealing zone assembly.PLoS One2e179 • • • • 25. Shyu JF, Shih C, Tseng CY, Lin CH, Sun DT, et al. (2007) Calcitonin induces podosome disassembly and detachment of osteoclasts by modulating Pyk2 and Src activities. Bone 40: 1329–1342.JF ShyuC. ShihCY TsengCH LinDT Sun2007Calcitonin induces podosome disassembly and detachment of osteoclasts by modulating Pyk2 and Src activities.Bone • • • • 26. Nakamura I, Kadono Y, Takayanagi H, Jimi E, Miyazaki T, et al. (2002) IL-1 regulates cytoskeletal organization in osteoclasts via TNF receptor-associated factor 6/c-Src complex. J Immunol 168: 5103–5109.I. Miyazaki2002IL-1 regulates cytoskeletal organization in osteoclasts via TNF receptor-associated factor 6/c-Src complex.J Immunol9 • • • • 27. Kirstein B, Chambers TJ, Fuller K (2006) Secretion of tartrate-resistant acid phosphatase by osteoclasts correlates with resorptive behavior. J Cell Biochem 98: 1085–1094.B. KirsteinTJ ChambersK. Fuller2006Secretion of tartrate-resistant acid phosphatase by osteoclasts correlates with resorptive behavior.J Cell Biochem • • • • 28. Fuller K, Kirstein B, Chambers TJ (2006) Murine osteoclast formation and function: differential regulation by humoral agents. Endocrinology 147: 1979–1985.K. KirsteinTJ Chambers2006Murine osteoclast formation and function: differential regulation by humoral agents.Endocrinology5 • • • • 29. Baharloo B, Textor M, Brunette DM (2005) Substratum roughness alters the growth, area, and focal adhesions of epithelial cells, and their proximity to titanium surfaces. J Biomed Mater Res A 74: 12–22.B. TextorDM Brunette2005Substratum roughness alters the growth, area, and focal adhesions of epithelial cells, and their proximity to titanium surfaces.J Biomed Mater Res A741222 • • • • 30. Grossner-Schreiber B, Herzog M, Hedderich J, Duck A, Hannig M, et al. (2006) Focal adhesion contact formation by fibroblasts cultured on surface-modified dental implants: an in vitro study. Clin Oral Implants Res 17: 736–745.B. Hannig2006Focal adhesion contact formation by fibroblasts cultured on surface-modified dental implants: an in vitro study.Clin Oral Implants Res17736745 • • • • 31. McSheehy PM, Chambers TJ (1986) Osteoblastic cells mediate osteoclastic responsiveness to parathyroid hormone. Endocrinology 118: 824–828.PM McSheehyTJ Chambers1986Osteoblastic cells mediate osteoclastic responsiveness to parathyroid hormone.Endocrinology118824828 • • • • 32. Holtrop ME, Raisz LG, Simmons HA (1974) The effects of parathyroid hormone, colchicine, and calcitonin on the ultrastructure and the activity of osteoclasts in organ culture. J Cell Biol 60: 346–355.ME HoltropLG RaiszHA Simmons1974The effects of parathyroid hormone, colchicine, and calcitonin on the ultrastructure and the activity of osteoclasts in organ culture.J Cell Biol60346355 • • • • 33. Chambers TJ, Revell PA, Fuller K, Athanasou NA (1984) Resorption of bone by isolated rabbit osteoclasts. J Cell Sci 66: 383–399.TJ ChambersPA RevellK. FullerNA Athanasou1984Resorption of bone by isolated rabbit osteoclasts.J Cell Sci66383399 • • • • 34. Charo IF, Nannizzi L, Smith JW, Cheresh DA (1990) The vitronectin receptor alpha v beta 3 binds fibronectin and acts in concert with alpha 5 beta 1 in promoting cellular attachment and spreading on fibronectin. J Cell Biol 111: 2795–2800.IF CharoL. NannizziJW SmithDA Cheresh1990The vitronectin receptor alpha v beta 3 binds fibronectin and acts in concert with alpha 5 beta 1 in promoting cellular attachment and spreading on fibronectin.J Cell Biol0 • • • • 35. Orr AW, Ginsberg MH, Shattil SJ, Deckmyn H, Schwartz MA (2006) Matrix-specific suppression of integrin activation in shear stress signaling. Mol Biol Cell 17: 4686–4697.AW OrrMH GinsbergSJ ShattilH. DeckmynMA Schwartz2006Matrix-specific suppression of integrin activation in shear stress signaling.Mol Biol Cell • • • • 36. Lakkakorpi PT, Helfrich MH, Horton MA, Vaananen HK (1993) Spatial organization of microfilaments and vitronectin receptor, alpha v beta 3, in osteoclasts. A study using confocal laser scanning microscopy. J Cell Sci 104(Pt 3): 663–670.PT LakkakorpiMH HelfrichMA HortonHK Vaananen1993Spatial organization of microfilaments and vitronectin receptor, alpha v beta 3, in osteoclasts. A study using confocal laser scanning microscopy.J Cell Sci104Pt 3663670 • • • • 37. Fuller K, Owens JM, Jagger CJ, Wilson A, Moss R, et al. (1993) Macrophage colony-stimulating factor stimulates survival and chemotactic behavior in isolated osteoclasts. J Exp Med 178: 1733–1744.K. FullerJM OwensCJ JaggerA. Moss1993Macrophage colony-stimulating factor stimulates survival and chemotactic behavior in isolated osteoclasts.J Exp Med4 • • • • 38. Huang HH, Ho CT, Lee TH, Lee TL, Liao KK, et al. (2004) Effect of surface roughness of ground titanium on initial cell adhesion. Biomol Eng 21: 93–97.HH HuangCT HoTH LeeTL LeeKK Liao2004Effect of surface roughness of ground titanium on initial cell adhesion.Biomol Eng219397 • • • • 39. Hughes DE, Salter DM, Dedhar S, Simpson R (1993) Integrin expression in human bone. J Bone Miner Res 8: 527–533.DE HughesDM SalterS. Simpson1993Integrin expression in human bone.J Bone Miner Res8527533 • • • • 40. Lowe C, Yoneda T, Boyce BF, Chen H, Mundy GR, et al. (1993) Osteopetrosis in Src-Deficient Mice Is Due to an Autonomous Defect of Osteoclasts. Proceedings of the National Academy of Sciences of the United States of America 90: 4485–4489.C. YonedaBF BoyceH. ChenGR Mundy1993Osteopetrosis in Src-Deficient Mice Is Due to an Autonomous Defect of Osteoclasts.Proceedings of the National Academy of Sciences of the United States of America • • • • 41. Boyce BF, Yoneda T, Lowe C, Soriano P, Mundy GR (1992) Requirement of Pp60c-Src Expression for Osteoclasts to Form Ruffled Borders and Resorb Bone in Mice. Journal of Clinical Investigation 90: 1622–1627.BF BoyceT. SorianoGR Mundy1992Requirement of Pp60c-Src Expression for Osteoclasts to Form Ruffled Borders and Resorb Bone in Mice.Journal of Clinical Investigation • • • • 42. Horne WC, Neff L, Chatterjee D, Lomri A, Levy JB, et al. (1992) Osteoclasts Express High-Levels of Pp60(C-Src) in Association with Intracellular Membranes. Journal of Cell Biology 119: 1003–1013.WC HorneL. LomriJB Levy1992Osteoclasts Express High-Levels of Pp60(C-Src) in Association with Intracellular Membranes.Journal of Cell Biology3 • • • • 43. Arias-Salgado EG, Lizano S, Sarkar S, Brugge JS, Ginsberg MH, et al. (2003) Src kinase activation by direct interaction with the integrin beta cytoplasmic domain. Proceedings of the National Academy of Sciences of the United States of America 100: 2.EG Arias-SalgadoS. SarkarJS BruggeMH Ginsberg2003Src kinase activation by direct interaction with the integrin beta cytoplasmic domain.Proceedings of the National Academy of Sciences of the United States of America302 • • • • 44. Felsenfeld DP, Schwartzberg PL, Venegas A, Tse R, Sheetz MP (1999) Selective regulation of integrin-cytoskeleton interactions by the tyrosine kinase Src. Nature Cell Biology 1: 200–206.DP FelsenfeldPL SchwartzbergA. TseMP Sheetz1999Selective regulation of integrin-cytoskeleton interactions by the tyrosine kinase Src.Nature Cell Biology1200206 • • • • 45. Geblinger D, Geiger B, Addadi L (2009) Surface-induced regulation of podosome organization and dynamics in cultured osteoclasts. Chembiochem 10: 158–165.D. Addadi2009Surface-induced regulation of podosome organization and dynamics in cultured osteoclasts.Chembiochem10158165 • • • • 46. Boskey AL, Maresca M, Ullrich W, Doty SB, Butler WT, et al. (1993) Osteopontin-Hydroxyapatite Interactions in-Vitro - Inhibition of Hydroxyapatite Formation and Growth in a Gelatin-Gel. Bone and Mineral 22: 147–159.AL BoskeyM. UllrichSB DotyWT Butler1993Osteopontin-Hydroxyapatite Interactions in-Vitro - Inhibition of Hydroxyapatite Formation and Growth in a Gelatin-Gel.Bone and Mineral22147159 • • • • 47. Oldberg A, Franzen A, Heinegard D (1986) Cloning and Sequence-Analysis of Rat Bone Sialoprotein (Osteopontin) Cdna Reveals an Arg-Gly-Asp Cell-Binding Sequence. Proceedings of the National Academy of Sciences of the United States of America 83: 8819–8823.A. Heinegard1986Cloning and Sequence-Analysis of Rat Bone Sialoprotein (Osteopontin) Cdna Reveals an Arg-Gly-Asp Cell-Binding Sequence.Proceedings of the National Academy of Sciences of the United States of America • • • • 48. Huq NL, Cross KJ, Reynolds EC (2000) Molecular modelling of a multiphosphorylated sequence motif bound to hydroxyapatite surfaces. Journal of Molecular Modeling 6: 35–47.NL HuqKJ CrossEC Reynolds2000Molecular modelling of a multiphosphorylated sequence motif bound to hydroxyapatite surfaces.Journal of Molecular Modeling63547 • • • • 49. Robey PG, Young MF, Fisher LW, McClain TD (1989) Thrombospondin is an osteoblast-derived component of mineralized extracellular matrix. J Cell Biol 108: 719–727.PG RobeyMF YoungLW FisherTD McClain1989Thrombospondin is an osteoblast-derived component of mineralized extracellular matrix.J Cell Biol108719727 • • • • 50. Chambers TJ, Fuller K (1985) Bone-Cells Predispose Bone Surfaces to Resorption by Exposure of Mineral to Osteoclastic Contact. Journal of Cell Science 76: 155–165.TJ ChambersK. Fuller1985Bone-Cells Predispose Bone Surfaces to Resorption by Exposure of Mineral to Osteoclastic Contact.Journal of Cell Science76155165 • • • • 51. Bianco P, Fisher LW, Young MF, Termine JD, Robey PG (1991) Expression of Bone Sialoprotein (Bsp) in Developing Human Tissues. Calcified Tissue International 49: 421–426.P. BiancoLW FisherMF YoungJD TerminePG Robey1991Expression of Bone Sialoprotein (Bsp) in Developing Human Tissues.Calcified Tissue International49421426 • • • • 52. Dodds RA, Connor JR, James IE, Rykaczewski EL, Appelbaum E, et al. (1995) Human osteoclasts, not osteoblasts, deposit osteopontin onto resorption surfaces: an in vitro and ex vivo study of remodeling bone. J Bone Miner Res 10: 1666–1680.RA DoddsJR ConnorIE JamesEL RykaczewskiE. Appelbaum1995Human osteoclasts, not osteoblasts, deposit osteopontin onto resorption surfaces: an in vitro and ex vivo study of remodeling bone.J Bone Miner Res • • • • 53. Arai N, Ohya K, Kasugai S, Shimokawa H, Ohida S, et al. (1995) Expression of Bone Sialoprotein Messenger-Rna during Bone-Formation and Resorption Induced by Colchicine in Rat Tibial Bone-Marrow Cavity. Journal of Bone and Mineral Research 10: 1209–1217.N. Ohida1995Expression of Bone Sialoprotein Messenger-Rna during Bone-Formation and Resorption Induced by Colchicine in Rat Tibial Bone-Marrow Cavity.Journal of Bone and Mineral Research • • • • 54. Tezuka K, Sato T, Kamioka H, Nijweide PJ, Tanaka K, et al. (1992) Identification of Osteopontin and Cathepsin L-Like Molecule in Isolated Rabbit Osteoclasts. Journal of Bone and Mineral Research 7: S130–S130.K. KamiokaPJ NijweideK. Tanaka1992Identification of Osteopontin and Cathepsin L-Like Molecule in Isolated Rabbit Osteoclasts.Journal of Bone and Mineral Research7S130S130 • • • • 55. Boyd NA, Bradwell AR, Thompson RA (1993) Quantitation of vitronectin in serum: evaluation of its usefulness in routine clinical practice. J Clin Pathol 46: 1042–1045.NA BoydAR BradwellRA Thompson1993Quantitation of vitronectin in serum: evaluation of its usefulness in routine clinical practice.J Clin Pathol • • • • 56. Shaffer MC, Foley TP, Barnes DW (1984) Quantitation of Spreading Factor in Human Biologic Fluids. Journal of Laboratory and Clinical Medicine 103: 783–791.MC ShafferTP FoleyDW Barnes1984Quantitation of Spreading Factor in Human Biologic Fluids.Journal of Laboratory and Clinical Medicine103783791 • • • • 57. Chambers TJ, Hall TJ (1991) Cellular and molecular mechanisms in the regulation and function of osteoclasts. Vitam Horm 46: 41–86.TJ ChambersTJ Hall1991Cellular and molecular mechanisms in the regulation and function of osteoclasts.Vitam Horm464186 • • • • 58. Duong LT, Lakkakorpi P, Nakamura I, Rodan GA (2000) Integrins and signaling in osteoclast function. Matrix Biol 19: 97–105.LT DuongP. NakamuraGA Rodan2000Integrins and signaling in osteoclast function.Matrix Biol1997105 • • •. [] An osteoclast (from the Greek words for 'bone' (ὀστέον), and 'broken' (κλαστός)) is a type of that breaks down. This function is critical in the maintenance, repair, and of of the. The osteoclast disassembles and digests the composite of hydrated protein and mineral at a molecular level by secreting acid and a, a process known as. This process also helps regulate the level of blood. An odontoclast (/odontoclast/; o-don´to-klast) is an osteoclast associated with absorption of the roots of. Illustrated cross-section of an activated osteoclast An osteoclast is a large multinucleated cell and human osteoclasts on bone typically have five nuclei and are about 150-200 µm in diameter. When osteoclast-inducing cytokines are used to convert to osteoclasts, very large cells that may reach 100 µm in diameter occur. These may have dozens of nuclei, and typically express major osteoclast proteins but have significant differences from cells in living bone because of the not-natural substrate. The size of the multinucleated assembled osteoclast allows it to focus the ion transport, protein secretory and vesicular transport capabilities of many macrophages on a localized area of bone. Location [ ] In bone, osteoclasts are found in pits in the bone surface which are called resorption bays,. Osteoclasts are characterized by a cytoplasm with a homogeneous, 'foamy' appearance. This appearance is due to a high concentration of and. These vacuoles include filled with. This permits characterization of osteoclasts by their staining for high of (TRAP) and. Osteoclast rough endoplasmic reticulum is sparse, and the Golgi complex is extensive. At a site of active bone resorption, the osteoclast forms a specialized, the 'ruffled border,' that opposes the surface of the bone tissue. This extensively folded or ruffled border facilitates bone removal by dramatically increasing the cell surface for secretion and uptake of the resorption compartment contents and is a morphologic characteristic of an osteoclast that is actively resorbing bone. Development [ ] Since their discovery in 1873 there has been considerable debate about their origin. Three theories were dominant: from 1949 to 1970 the connective tissue origin was popular, which stated that osteoclasts and are of the same lineage, and osteoblasts fuse together to form osteoclasts. After years of controversy it is now clear that these cells develop from the self fusion of macrophages. It was in the beginning of 1980 that the phagocytic system was recognized as precursor of osteoclasts. Osteoclast formation requires the presence of (receptor activator of nuclear factor κβ ligand) and. These membrane-bound proteins are produced by neighbouring and, thus requiring direct contact between these cells and osteoclast. M-CSF acts through its receptor on the osteoclast, c-fms (colony-stimulating factor 1 receptor), a transmembrane -receptor, leading to activation of tyrosine kinase Src. Both of these molecules are necessary for osteoclastogenesis and are widely involved in the of monocyte/macrophage derived cells. RANKL is a member of the tumour necrosis family (), and is essential in osteoclastogenesis. RANKL knockout mice exhibit a phenotype of and defects of tooth eruption, along with an absence or deficiency of osteoclasts. RANKL activates NF-κβ (nuclear factor-κβ) and NFATc1 (nuclear factor of activated t cells, cytoplasmic, calcineurin-dependent 1) through. NF-κβ activation is stimulated almost immediately after RANKL-RANK interaction occurs and is not upregulated. NFATc1 stimulation, however, begins ~24–48 hours after binding occurs and its expression has been shown to be RANKL dependent. Osteoclast differentiation is inhibited by (OPG), which is produced by osteoblasts and binds to RANKL thereby preventing interaction with RANK. It may be important to note that while osteoclasts are derived from the hematopoietic lineage, osteoblasts are derived from mesenchymal stem cells. Function [ ] Once activated, osteoclasts move to areas of microfracture in the bone. Osteoclasts lie in a small cavity called Howship's lacunae, formed from the digestion of the underlying bone. The sealing zone is the attachment of the osteoclast's to the underlying bone. Sealing zones are bounded by belts of specialized adhesion structures called. Attachment to the bone matrix is facilitated by integrin receptors, such as αvβ3, via the specific Arg-Gly-Asp in bone matrix proteins, such as. The osteoclast releases hydrogen ions through the action of ( + → − + +) through the ruffled border into the resorptive cavity, acidifying and aiding dissolution of the mineralized bone into, H 3PO 4, H 2CO 3, water and other substances. Dysfunction of the carbonic anhydrase has been documented to cause some forms of osteopetrosis. Hydrogen ions are pumped against a high concentration gradient by, specifically a unique. This enzyme has been targeted in the prevention of. In addition, several, such as members of the and matrix metalloprotease (MMP) groups, are released to digest the organic components of the matrix. These enzymes are released into the compartment. Of these hydrolytic enzymes, cathepsin K is of most importance. Cathepsin K and other cathepsins [ ] is a collagenolytic, -like, that is mainly expressed in osteoclasts, and is secreted into the resorptive pit. Cathepsin K is the major involved in the degradation of type I collagen and other noncollagenous proteins. Mutations in the cathepsin K gene are associated with, a hereditary disease, characterised by a lack of functional cathepsin K expression. Knockout studies of cathepsin K in mice lead to an osteopetrotic phenotype, which, is partially compensated by increased expression of proteases other that cathepsin K and enhanced osteoclastogenesis. Cathepsin K has an optimal enzymatic activity in acidic conditions. It is synthesized as a proenzyme with a molecular weight of 37kDa, and upon activation by autocatalytic cleavage, is transformed into the mature, active form with a molecular weight of ~27kDa. Upon polarization of the osteoclast over the site of resorption, cathepsin K is secreted from the ruffled border into the resorptive pit. Cathepsin K transmigrates across the ruffled border by intercellular vesicles and is then released by the functional secretory domain. Within these intercellular vesicles, cathepsin K, along with generated by, further degrades the bone extracellular matrix. Several other cathepsins are expressed in osteoclasts including,, D, E, G, and L. The function of these and is generally unknown within bone, and they are expressed at much lower levels than cathepsin K. Studies on cathepsin L have been mixed, with a report of reduced in and cathepsin L knockout mice compared to wild-type and another report finding no skeletal abnormalities. Matrix metalloproteinases [ ] The (MMPs) comprise a family of more than 20 zinc-dependent endopeptidases. The role of matrix metalloproteinases (MMPs) in osteoclast biology is ill-defined, but in other tissue they have been linked with tumor promoting activities, such as activation of and are required for tumor metastasis and angiogenesis. MMP-9 is associated with the bone microenvironment. It is expressed by osteoclasts, and is known to be required for osteoclast and is a powerful gelatinase. Transgenic mice lacking MMP-9 develop defects in bone development, intraosseous, and fracture repair. MMP-13 is believed to be involved in bone resorption and in osteoclast differentiation, as knockout mice revealed decreased osteoclast numbers, osteopetrosis, and decreased bone resorption. MMPs expressed by the osteoclast include MMP-9, -10, -12, and -14. Apart from MMP-9, little is known about their relevance to the osteoclast, however, high levels of MMP-14 are found at the sealing zone. Osteoclast Physiology [ ] In the 1980s and 90s the physiology of typical osteoclasts was studied in detail. With the isolation of the ruffled border, ion transport across it was studied directly in biochemical detail. Energy-dependent acid transport was verified and the postulated proton pump purified. With the successful culture of osteoclasts, it became apparent that they are organized to support the massive transport of protons for acidification of the resorption compartment and solubilization of the bone mineral. This includes ruffled border Cl − permeability to control membrane potential and basolateral Cl −/HCO 3 − exchange to maintain cytosolic pH in physiologically acceptable ranges. > The effectiveness of its ion secretion depends upon the osteoclast forming an effective seal around the resorption compartment. The positioning of this 'sealing zone' appears to be mediated by integrins expressed on the osteoclast surface. With the sealing zone in place, the multinucleated osteoclast reorganizes itself. Developing the highly invaginated ruffled membrane apposing the resorption compartment allows massive secretory activity. In addition, it permits the vesicular of the mineral and degraded collagen from the ruffled border to the free membrane of the cell, and its release into the extracellular compartment. This activity completes the bone resorption, and both the mineral components and collagen fragments are released to the general circulation. Regulation [ ] Osteoclasts are regulated by several, including (PTH) from the parathyroid gland, from the thyroid gland, and (IL-6). This last hormone,, is one of the factors in the disease, which is an imbalance between bone resorption and bone formation. Osteoclast activity is also mediated by the interaction of two molecules produced by osteoblasts, namely and. Note that these molecules also regulate differentiation of the osteoclast. Odontoclast [ ] An odontoclast (/odontoclast/; o-don´to-klast) is an osteoclast associated with absorption of the roots of. Alternate use of term [ ] An osteoclast can also be an instrument used to fracture and reset bones (the origin is Greek osteon: bone and klastos: broken). To avoid confusion, the cell was originally termed osotoclast. When the surgical instrument went out of use, the cell became known by its present name. Clinical significance [ ] Giant osteoclasts can occur in some diseases, including and toxicity. History [ ] Osteoclasts were discovered by Kolliker in 1873. References [ ]. 
Almost a year ago, Sanyo released its messaging phone for. Though we appreciated its spacious keyboard and call quality, we weren't so impressed by the exterior touch controls and tiny display. 
Sure, the design was attractive and original, but it just wasn't for us. Now, just over 11 months later, Sanyo and Sprint are giving the design another go with the Innuendo SCP-6780 (no, we don't get the 'Innuendo' name, either). It's slimmer and less angular, but like its predecessor it's not always easy to use. You can get it for $49.99 with a two-year contract and after a $50 mail-in rebate. Design As mentioned, the Innuendo closely resembles the earlier Incognito. Apr 10, 2015 Sanyo Innuendo by Kyocera for Sprint. MANUALLY FLASH & ACTIVATE SPRINT S5 TO BOOST 4G LTE 2ND VIDEO - Duration: 3:17. Genius wireless pro 3,687 views. ACTIVATE ONLINE OR BY PHONE ∙To activate online, visit: www.boostmobile.com/activate ∙To activate by phone, call. 
Indeed, you'll see the same exterior features and flip-open design. It's also about the same size as the Incognito (4.2 inches long by 2.2 inches wide by 0.6 inch thick), but the curved ends give it a trim and more aerodynamic profile. The Innuendo has a more attractive blue color, but its glossy surface also shows smudges and fingerprints way too easily. We like that it catches the light--and you can use it to check your teeth before a date--but the fingerprints are distracting and unappealing. The Innuendo features touch controls and a glossy exterior. The exterior touch controls are similar as well, though Sanyo tweaked the design slightly and added a dedicated speakerphone control. Otherwise, you'll find the standard 12-digit keypad, a back key, the Talk and End buttons, four directional arrows, and a central OK button. Here again, the arrangement is spacious, but you can't dial by feel and the surface is slick. Vibration feedback helps a bit, but not much. What's more, we remain dissatisfied with the smaller external display. Not only is it monochrome, so it won't show photo caller ID, but also you can access only a few menu options. In contrast, the Innuendo mixes up the remaining external controls just a bit. The Micro-USB charger port and small volume rocker remain on the right spine, but the microSD card slot is now inconveniently located behind the battery. The camera lens sits on the rear side next to a speaker and on the top of the device are a 2.5mm headset jack and the power control. The hinge runs the length of the Innuendo's left spine. It's a tad bulky, but the trade-off is a sturdy construction that feels solid (3.4 ounces). The mechanism is neither too loose nor too stiff, and the phone clicks into place on either end. The Innuendo opens almost a full 180 degrees, albeit with a slight S curve. Unless it's opened completely, it wobbles slightly when resting on a table. The internal display measures 2.8 inches (the Incognito has a 2.6-inch screen) and supports 262,000 colors. Its resolution (400x240 pixels) is slightly higher than the Incognito, which means you'll have a rich viewing experience. The display has a landscape orientation, and Sprint's OneClick interface offers customizable shortcuts to favorite features. The Innuendo has a spacious, comfortable keyboard. Below the hinge are the two soft keys. As they're not directly under the corresponding command on the screen, the arrangement may not be intuitive at first. The keyboard has just three rows, which means letters share space with numbers and symbols, but the keys are large and spaced far apart. We could dial and text quickly without any issues, and we appreciate that the buttons aren't completely flush. You'll also find a navigation toggle, Talk and End/power buttons, a back control, a camera shutter, a speakerphone control, a key for accessing emoticons, a messaging shortcut, and a spacious, well-positioned space bar. Features With the exception of a higher-resolution camera, the Innuendo's feature set is almost unchanged from the Incognito. As such, portions of this section are taken from the. The Innuendo's 600-contact phone book has room in each entry for multiple phone numbers, an e-mail address, a street address, an instant-messaging ID, a URL, and notes. You also can organize callers into groups and pair them with a photos and one of 39 (72-chord) polyphonic ringtones. Sprint offers a wireless backup service for your contacts in case you lose your phone. The new action-thriller from the award-winning team at Infinity Ward, the creators of the Call of Duty series, delivers the most intense and cinematic action experience ever. Call of Duty 4: Modern Warfare arms gamers with an arsenal of advanced and powerful modern day firepower and transports them to the most treacherous hotspots around the globe to take on a rogue enemy group threatening the world. As both a U.S Marine and British S.A.S. Soldier fighting through an unfolding story full of twists and turns, players use sophisticated technology, superior firepower and coordinated land and air strikes on a battlefield where speed, accuracy and communication are essential to victory. The epic title also delivers an added depth of multiplayer action providing online fans an all-new community of persistence, addictive and customizable gameplay. • Key features. • Authentic Advanced Weaponry - Featuring an available arsenal of more than 70 new and authentic weapons and gear from assault rifles with laser sites, claymore mines,.50 caliber sniper rifles, and M-249 SAW machine guns. With accessories like night-vision goggles and ghillie suits, for maximum concealment, Call of Duty 4: Modern Warfare has players locked and loaded to accomplish the mission. • Coordinated Assault and Support - Delivering the most visceral action thriller ever, the title covers modern battle from the soldier to the satellite, where the need for air support is critical to success. The adrenaline rush deployment enlists gamers to fast-rope from tactical helicopters, ride in an armada of attack choppers, utilize jets to remove enemy strongholds and even engage hostiles from thousands of feet above the ground inside a state of the art aerial gunship. • Cinematic Quality Graphics and Sound - Featuring stunning next-generation graphics, players will be drawn into the cinematic intensity of Call of Duty 4: Modern Warfare. Just checking to see if it is possible to register my retail CD key with Steam and then download the client? I did with with my retail UT3 and Dark. May 27, 2009 Just checking to see if it is possible to register my retail CD key with Steam and then download the client? I did with with my retail UT3 and Dark.   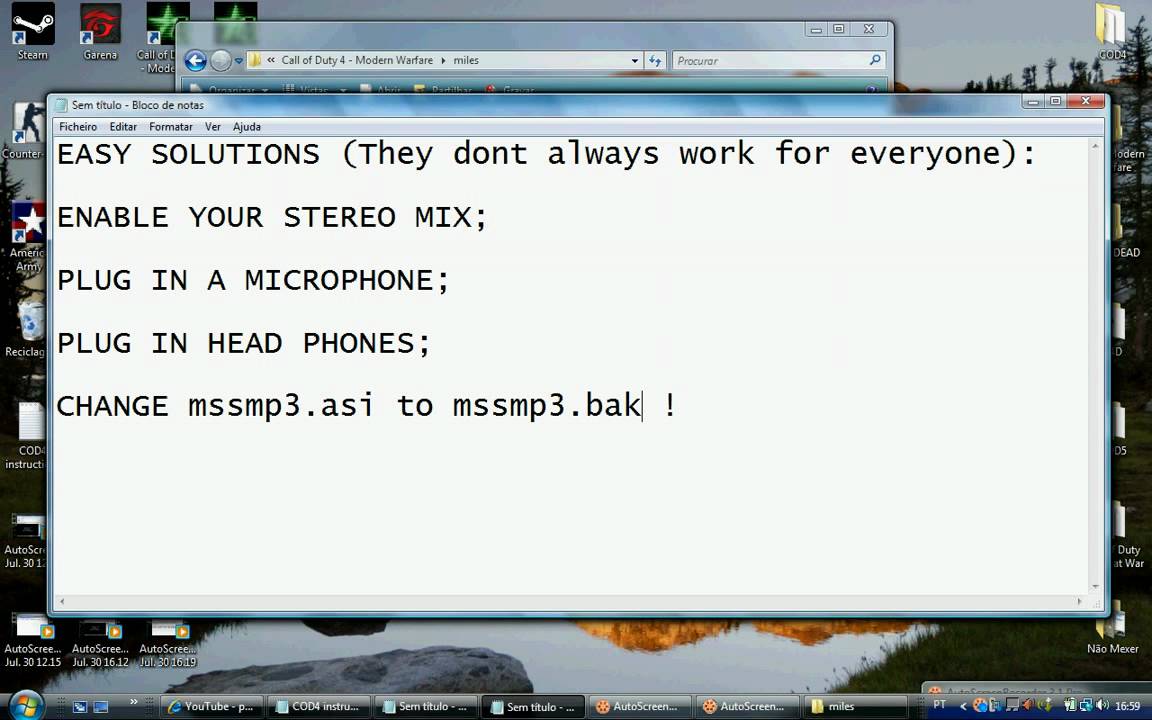 
Amazing special effects, including realistic depth of field, rim-lighting, character self-shadowing, texture streaming as well as physics-enabled effects will enlist players into the most photo-realistic gaming experience. Combine the lifelike graphics and the realistic battle chatter with the Call of Duty award-winning sound design and players will face battle as they have never before. • Unparalleled Depth to Multiplayer - Multiplayer builds from the success of Call of Duty 2 delivering a persistent online experience for greater community interaction. Featuring create-a-class options allowing players to customize gear that is best suited for play, to experience points enabling unlockables and perks, all the way to matchmaking and leaderboards for the latest in tracking, Call of Duty 4: Modern Warfare's multiplayer is set to deliver easily accessible and addictive online play for all. Activate Vodafone 4G internet APN Settings 2016 for Australia, India, UK, Egypt, Germany, Turkey, Italy, Portugal, Netherland, Romania, Spain, New Zealand: Hi there, are you searching on Google for a trick to get your Vodafone 4G Settings for all Countries then you have landed on the right place here in this article explain How to Activate Vodafone 4G LTE Internet, APN, MMS Settings for All Mobiles like Samsung Galaxy s4, Nokia Lumia, i PHONE, Windows phone, Android Phones, Sony Xperia, HTC, Blackberry, Unlocked iPhone, Unlocked Phone. Now let’s see how to do VODAFONE LTE 4G Mobile internet Settings trics for different countries. What Is Meaning Of vodafone 4g internet? Lte stands for ‘long term evolution’ – it is a radio access technology to deliver very high speeds. 4g is a standard in mobile network technology and stands for ‘fourth generation’ – the latest after 2g and 3g which gives you a faster and rich internet experience. With 4g lte hi-speeds, you can notice a visible difference while downloading files, doing video-chat, playing multiplayer games or while viewing hd videos online. 
Disclaimer All content, information and/or links provided within this website or the site linking to this disclaimer, are offered only as general information. This is to further inform the subscribers /customers / users that by clicking on the link, you will enter the website of a third party which is not owned, controlled or endorsed by Vodafone India Limited and its affiliates ('Vodafone'). Such information is provided only for the convenience of the subscribers / customers / users and Vodafone does not undertake any liability or responsibility for the details, accuracy, completeness or correct sequence of any content or information provided in the linked site. Neither Vodafone nor any of its agents or licensors shall be liable for any direct, indirect, incidental, special or consequential loss or damages (including, without limitation for loss of profit, business opportunity or loss of goodwill) whatsoever, whether in contract, tort, misrepresentation or otherwise arising from the use of the linked site. The information available on the website is subject to change, update, revision, verification and amendment without notice and such information may change materially. All content, information and/or links provided within this website or the site linking to this disclaimer, are offered only as general information. This is to further inform the subscribers /customers / users that by clicking on the link, you will enter the website of a third party which is not owned, controlled or endorsed by Vodafone India Limited and its affiliates ('Vodafone'). Such information is provided only for the convenience of the subscribers / customers / users and Vodafone does not undertake any liability or responsibility for the details, accuracy, completeness or correct sequence of any content or information provided in the linked site. Neither Vodafone nor any of its agents or licensors shall be liable for any direct, indirect, incidental, special or consequential loss or damages (including, without limitation for loss of profit, business opportunity or loss of goodwill) whatsoever, whether in contract, tort, misrepresentation or otherwise arising from the use of the linked site. The information available on the website is subject to change, update, revision, verification and amendment without notice and such information may change materially.   
Blackberry Apn Settings For Vodafone Egypt. Here's how activate your globe gprs/mms/3g 560 x 354 52 kB jpeg, Vodafone Egypt Blackberry Inter Service. Vodafone Egypt 4G LTE 3G Internet and MMS APN Settings for Android Samsung Galaxy HTC Nexus Vodafone Egypt APN Settings for Android In your Android Smart. APN: internet.vodafone.net. Proxy: Not Set Port: Not Set Username: internet. Password: internet. Server: Not Set MMSC: Not Set MMS Proxy: Not Set. 
Welcome to Cricket! If you recently purchased a universal SIM card kit or device from a non-Cricket retailer such as Amazon, Target, or Walmart, use this easy step-by-step guide to activate your Cricket service. We'll walk you through creating a Cricket account, choosing a plan, and installing your SIM card. Get ready to enjoy Cricket's works-everywhere network! What You Need: • SIM card and device from a non-Cricket retailer OR Universal SIM card kit from a non-Cricket retailer and a GSM-compatible unlocked device • A copy of your current wireless bill (if you want to transfer your number) • A debit/credit card, Cricket Refill Card, or Service Payment Card to pay for your first month's service Let's get started! Visit cricketwireless.com/join and follow the step-by-step guide below. If you need help during the setup process, just with a friendly Cricket Support Advocate. First, you'll need to verify your ICCID and IMEI numbers • Your ICCID number is printed on the larger card (shaped like a credit card) that came with your SIM card. It can be up to 22 numbers long. If you bought a new device, the ICCID should also be printed on your box. • You can usually find your 15-digit IMEI number in your phone's settings: • On an iPhone, go to Settings > General > About and look for your IMEI. Before attempting to activate your AT&T SIM card, check whether your phone or device is already active. To do this, install the SIM card into the device and turn it. Define activate: to make active or more active: such as; to make (something, such as a molecule) reactive or more reactive — activate in a sentence. • On an Android, go to Settings > About Phone or About Tablet > Status and locate your IMEI. • Other ways to get your IMEI: • Look on the back of your phone (under the battery). • Look on your new device box. • Dial *#06# on your phone. • Enter both numbers and select Continue. Tell us what kind of customer you are • If you're an existing customer, you'll need to decide if you're adding a new line to your account or changing the device on an existing one. You can sign in to your account at this point. Choose your plan and features • Pick a rate plan and any optional add-on features; then select Continue. Choose your phone number • Choose Get a Cricket Number or Transfer Your Number (you'll complete the transfer later). 
Review your cart • Review your shopping cart and select Check Out. Create a Cricket account or verify your existing account • Enter your contact info and register for online account access. • Create a PIN and choose your security questions. • If you're already a customer, make sure your information is correct. Enter your payment info • Enter your billing address. • Select your payment method: Credit Card or Service Payment Card -or- Cricket Refill Card. You can also make a split payment using one or more Refill Cards in combination with a debit/credit card or Service Payment Card. • If you're paying with a debit/credit card or Service Payment Card, enter your card info. Agree to the terms and conditions, and select Confirm. • If you're paying with a Cricket Refill Card, enter the Refill Card number and select Validate. If you want to add another Refill Card, you can. • You can always add another Refill Card or a credit card by selecting Add or Edit. • If you're paying with a debit/credit card, you have the option to turn on Auto Pay. If not, you can always set up Auto Pay later in My Account or the myCricket app. • Review your info and select Continue. Transfer your current wireless number (optional) • Gather your current wireless account information. This includes your account number, account PIN or password, and the last four digits of the account holder's Social Security Number. Look on a recent bill or call you current wireless carrier if you need help. • Use the exact info that appears on your bill to avoid potential problems. • All fields may not be required to verify your number transfer, but fill out as many as you can. • The number transfer process will not begin until you activate your service. • Don't cancel your old wireless service. It will cancel automatically once the transfer is complete. Complete your order and activate • Review your info and make any changes. • Select Place Order to finish. • Print or save the confirmation number for your records (we'll also email you the details). • Make sure you select Activate from the receipt page. Please insert your SIM card first before you activate. See instructions below. • If we experience a delay in confirming your number transfer, you'll have to wait to activate. We'll email you as soon as we confirm the transfer with a link to activate your service. Choose your SIM card size (if you brought your own device): • To determine your SIM card size, remove your phone's back cover and battery. If you can't find the SIM card there, your phone may use a slide-out tray on the top or the side to hold the SIM card. You'll need the SIM tray tool (or a small paper clip) to open it. • Find the universal SIM card kit and identify your SIM card size. You may also want to review the Quick Start Guide that came with your package. Insert your SIM card and turn on your phone • Be sure you insert the SIM card into your device BEFORE you activate. • If you purchased a universal SIM card kit for a device you're bringing to Cricket, you'll need to choose your SIM card size (see above). • If your phone uses a nano SIM, you can insert the SIM directly into your phone's SIM card slot. • If your phone uses a micro SIM, you'll need one of the SIM tray adaptors that came with your SIM card kit. Place the SIM card into the tray that matches your SIM card size and insert it into your phone facing the correct way. • If you have questions, refer to your phone's user manual. You've joined the #CricketNation • After activation, your phone should start to work within minutes, usually immediately. You can use all the features included in your selected plan. • Depending on your phone, you may receive a series of messages and links from Cricket. Follow these to get the most out of your Cricket experience! Enhance your experience • Register for My Account online to enjoy easy online account management. Go to and register in two easy steps. You can make payments, change your plan or features, view your usage, and much more. • If you have a smartphone, we recommend downloading our helpful apps. • Get the app for your smartphone to make payments and view your usage right on your device. • Get the Cricket Wi-Fi app to help you save on data by connecting to free Wi-Fi networks wherever you go. How to activate your Roku account in Roku.com/link? • Open up your web browser in your laptop or computer. • Enter the www.roku.com/link in URL. • Once you see the web page, enter the Roku Activation code in it. • If you already have an existing account use that account to activate your device. • If you don’t have, you can create one. • For new users in order to create a new roku.com link account; • Click on “create new account” • Enter your first and last name • After entering your email address, create a password and verify it. This description of how to set up a Roku Video Streamer, will work for every current/recent model, and every future one they are likely to introduce in the near future. (Some older or obsolete models did not use HDMI as the primary style of video connection to convey the video signal to the TV, but the description which follows should be exactly what is needed for 98% of recent model installations. For the older ones; it is simply a matter of seeing which style of video connection is on both your TV and your Roku Streamer.) For Roku setup the streaming player needs 3 things – 2 inputs and 1 output: • Source of Internet (Network) – either wired or wireless • Source of Electrical Power • Output of Video 1) Internet – Wired connection is to be preferred for Roku activation; it is usually faster and is not subject to picking up interference from your neighbor’s wireless system. Some Roku devices have a Wired Internet input; some do not. Roku Stick and Roku Express generally use wireless, so skip the next part about Ethernet cable. If you have these, and you want to use the wired connection, plug the cable into the socket. If you do not, you will use wireless connection (“Wi-Fi”). Several steps from now, the device will show you a list of the wireless networks that it is able to receive a signal from, and you will choose the network you desire to use from the list, and enter the password for the network (if the network has a password; it should, otherwise anyone nearby can use your internet without permission). 2) Electrical Power – there are two styles of Power for Roku streaming devices. If you have a Roku TV to plug in the main power to the TV and the Roku streaming system inside the TV will automatically receive power also, and you can skip to step 3. For streaming devices: a) Wall Transformer that does not use USB – if your transformer has a non-detachable cord and looks somewhat like the following. Other end may have a right angle like the other end of the Express cable, but with a round plug instead of the pictured trapezoidal plug. So, plug the device end into your Stick or Express, and plug the other end into the wall power adapter for your Express, or for your Stick, use either the wall power adapter or any available USB port on the TV. If you plug into the USB port on the TV and there is a red light on your device, that means that the USB port on the TV is not delivering enough electrical current to the device for it to operate properly, and therefore you must use the wall power adapter instead. 3) Video output. HDMI cables carry both High-Definition video, and multi-channel audio simultaneously. With this in mind... A) Roku TV – all connections are internal and done already. B) Roku Streaming Stick – the male is built right into the stick, plug it into any available HDMI port on the television. C) Roku Express – connect HDMI cable male connectors on both ends, one to the HDMI on the Roku, the other to any available HDMI port on the TV. 4) Video Selection – use the TV’s remote control and/or the buttons on the TV, to ensure that the port you have plugged the Roku device into (or, the Roku part of a Roku TV), is the port that the TV will use to put the video onto the screen. 5) Insert the batteries into your remote.(There is a symbol inside the battery compartment in the remote that indicates the correct polarity). 6) The Roku logo will appear on your screen. If you do not get it then please check a) Is your player plugged into an electricity socket correctly? B) Double check that your TV is programmed for the same input of the HDMI cable and ensure that the cable is connected firmly on both ends of the player and TV. 7) Pair your remote a) To pair your remote open the battery door b) Find the button at the end and press for 3 seconds while aiming at the player, for Express and TV. For the Stick, because the stick will most often be behind the TV, such that the beam of invisible infra-red light which is used by the regular remote to communicate commands to the device, would not reach the Stick – so the Stick uses a radio signal that does not require the remote to be aimed at the Stick. 8) After selecting your language, the player will show the names of all wireless networks that it is able to communicate with. Choose your wireless network and enter the password. A) Success will be indicated by a green symbol; b) Failure with a red X. In most cases the password was entered incorrectly. (Note: The Password is case sensitive) 9) Player will attempt to automatically download software updates (this will take several minutes and then it will be installed automatically) After that, the player will restart. Then, the player will show on the TV screen, the Roku activation code which will be used to link your player to your Roku.com/link account. For help contact www.roku.com/link or reach out to Roku customer help. Measure 124.5 x 124.5 x 21.6 mm size larger than the Express and Express+. It is the best of the Roku Products. It provides everything that Roku ultra provides USB and Micro-SD slots for viewing your Personal Photos and your videos along with.MP4 playback. It also provides Dolby digital audio effect using HDMI or optical output. It also provides night listening mode. It remote supports private listening using headphone. The remote comes with the ability to connect the head phone. It comes with lost remote locator. This feature allows you to press a button in the box and this helps you locate the remote with an alert from the remote. It remote provides gaming buttons and voice search option this option is available in other devices only through the apps. It uses the RF technology instead of the IR blast. This means that the remote can be used even if the box is not in site. Roku Premiere+: Measure 124.5 x 124.5 x 21.6 mm size larger than the Express and Express+ Roku Premier+ is the one of the Roku top of the line models. It supports both Ethernet cable and dual band WiFi connectivity. It also provides Micro-SD card slots for viewing video and photos in your television. It is faster than Roku 1 which is the older model. Night listening mode available for this model. It is provides private listening option(The remote has the audio output jack. Headphones can be used in it for private listening) Roku Premiere+ Remote uses RF technology (The Box can be out of site and the remote can still be used). What does Roku provide as a streaming device? • It supports full 1080p HD videos and movies. • It provides a wide array of channels and TV shows • Remote control which includes settings and options • Streams through your wired or wireless network • Features a user friendly mobile app. • Headphones for private listening • Night listening mode (reduces loud noise and increases soft sounds) • Many different models such as- Roku (1-4), Roku Ultra, Roku premiere, Roku premiere+, Roku express, Roku express+ When using this device it is imperative to have high speed internet or your content quality may suffer. Roku is an electronic streaming device which enables you to stream your favorite movies, TV channels and shows to your TV via the internet. Even though most people know how to setup and activate their device, others face a bit of difficulty with the entire process.The problem that most users stumble upon relates to the www.roku.com/link during Roku Setup. In this site we are providing you with detailed information pertaining to your device on how to activate the device in roku.com/link website and will make your experience with Roku the best that it can be. Before we get into the details on how to setup and activate roku streaming player you need to have a thorough understanding of this device. Given above is the information relating to the this device and its setup. Roku offers a wide array of channels including both paid and free channels.It provides users with a HD video quality and uninterrupted streaming. The great thing about this device is that it is just a fraction of the cost when compared to going out and watching movies in theaters. You can sit back, relax and enjoy high quality entertainment from the comfort of your couch by activating this device at roku.com link. For more on this product check roku.com/link. Rokuactivationcode.com is an independent website that provides information for Roku streaming player issues. The website does not have specific association with “Roku” or its services. All information and images furnished on this website are for informational purpose only. We do not sell products or services of Roku in any form. We do not provide any warranty or services associated with the manufacturer. You should approach the manufacturer for the same. Users are advised to review our privacy policy before using our website. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
February 2018
Categories |
 RSS Feed
RSS Feed
